Researchers, clinicians, and commercial marketers all apply the term "peptide therapy" differently and for different purposes. Its meaning varies considerably depending on the context in which it appears. In peer-reviewed literature, the phrase describes the studied application of peptide compounds to influence specific biological mechanisms. In commercial settings, however, companies often attach it to unapproved products with claims that far exceed what available evidence actually supports.
This article explains what peptide therapy means, how peptides function at the molecular level, what the research literature has examined, and what regulatory and sourcing considerations matter most for researchers. All content here serves educational and laboratory research reference purposes only.
What Peptide Therapy Means
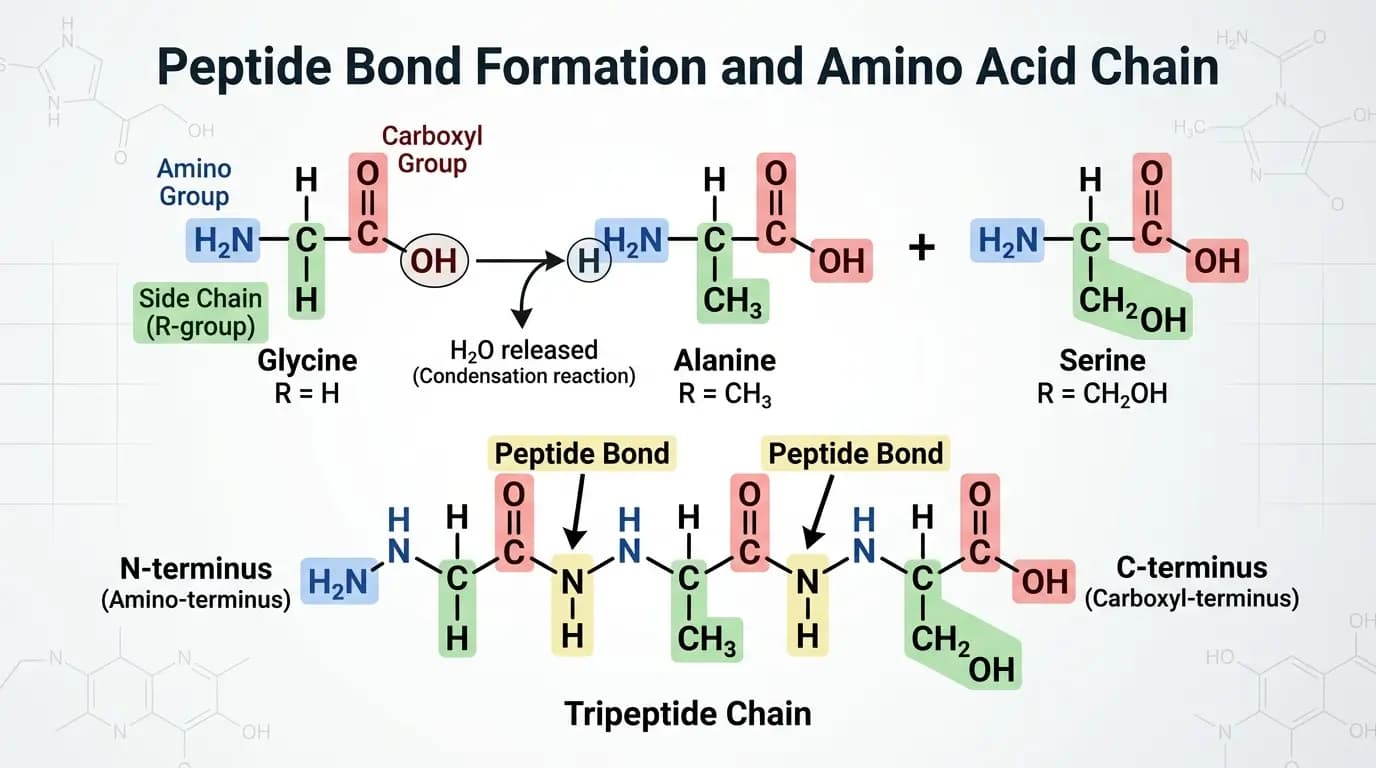
Peptide therapy, in its broadest sense, describes the application of peptide compounds to modulate specific biological pathways. The term appears in academic journals, in formally regulated medical contexts, and in commercial markets where companies apply it far less consistently. Understanding how the term functions across these different contexts is, therefore, essential before evaluating any specific claim made about it.
What Peptides Are and How They Function in the Body
Peptides are short chains of amino acids connected by peptide bonds. Structurally, they are smaller than proteins, generally containing between two and approximately fifty amino acids depending on classification conventions. In fact, the human body produces a wide range of peptides endogenously. Specifically, these include hormones, signaling molecules, and regulatory compounds that coordinate activity across organ systems and cellular networks.
Functionally, peptides operate primarily through receptor-mediated interactions. When a peptide encounters a complementary receptor on a cell surface, it binds and initiates or suppresses a cascade of intracellular signals. In turn, this mechanism of targeted binding is one key reason peptides attract sustained research interest. Furthermore, the structural selectivity of peptide-receptor interactions distinguishes them from many broader-acting pharmacological agents that researchers study in parallel research areas.
Well-documented natural peptides include insulin, glucagon, and oxytocin. Indeed, each of these compounds has a defined physiological role that extensive scientific study has established over many decades.
The Difference Between Natural Peptides and Synthetic Peptides
Natural peptides originate inside living organisms through biosynthesis pathways that the genome encodes. Complex feedback systems govern their production, activity, and regulation, and these systems respond dynamically to physiological states. Synthetic peptides, by contrast, emerge from controlled laboratory environments. Researchers produce them most commonly using solid-phase peptide synthesis (SPPS), a method that allows precise and reproducible construction of specific amino acid sequences.
In some cases, synthetic peptides mirror natural sequences exactly. In other cases, researchers modify them chemically to improve resistance to enzymatic degradation, alter receptor binding, or extend stability in biological fluids. Research applications favor synthetic peptides in many cases precisely because laboratories can confirm their purity and sequence analytically before use. Nevertheless, a synthetic peptide does not automatically replicate the behavior of its natural counterpart within the full complexity of a living biological system, and this distinction matters greatly when evaluating peptide therapy claims.
How Peptide Therapy Is Described to Work
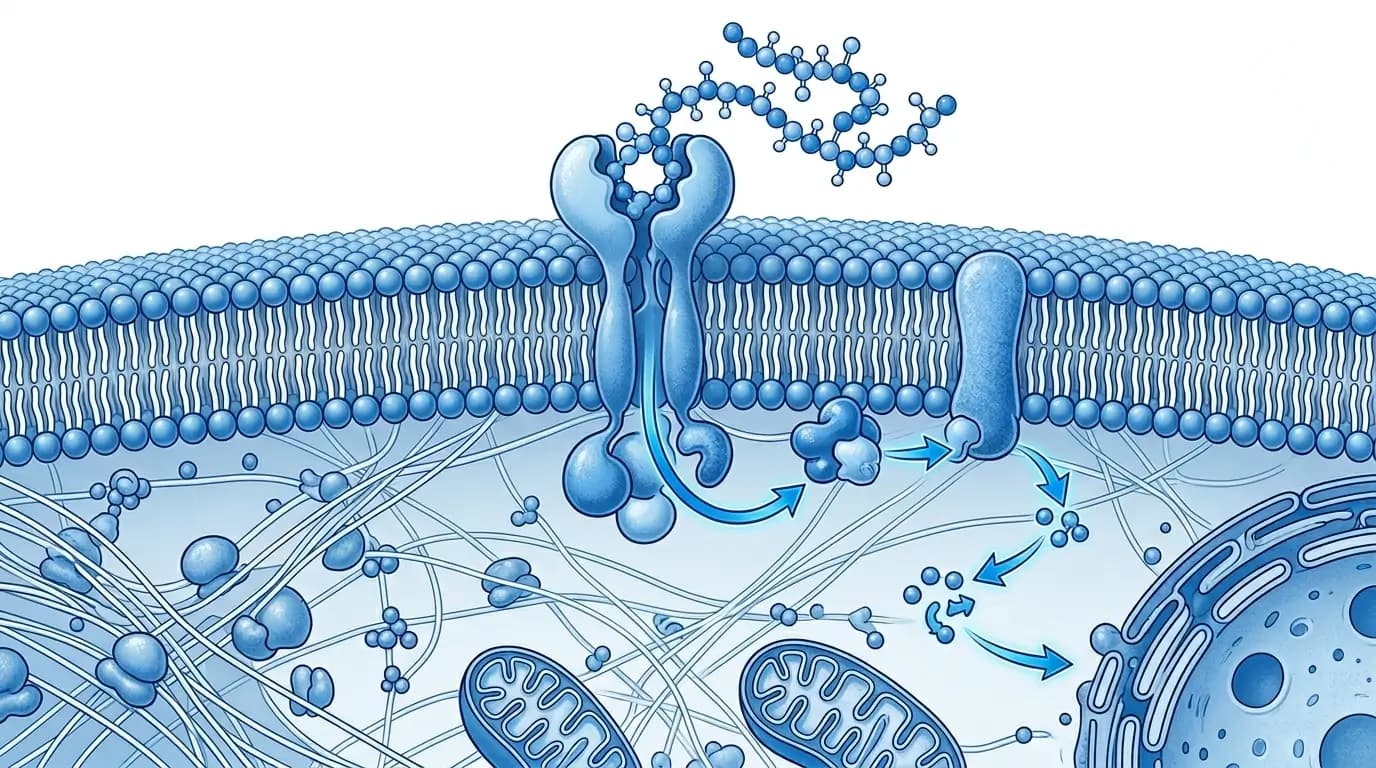
Researchers and proponents describe peptide therapy as functioning through specific, receptor-mediated biological interactions. In practice, the plausibility of this description for any given compound depends entirely on which peptide researchers are studying, what research model they have used, and how rigorously researchers have reviewed and replicated the available findings.
Cell Signaling, Receptors, and Targeted Biological Activity
Peptides exert their studied effects primarily through binding to specific receptors on the surface of target cells or within the intracellular environment. This binding activates or inhibits downstream signaling pathways, which can in turn influence processes such as protein synthesis, inflammatory signaling, gene expression, and metabolic regulation at the cellular level.
Notably, research publications frequently highlight the selectivity of this binding process as a structurally notable property of peptide compounds. Unlike many small-molecule agents, which interact with multiple receptor types simultaneously, certain peptides demonstrate narrow binding profiles in laboratory models. For instance, peer-reviewed studies in journals such as the Journal of Biological Chemistry and Peptides have examined receptor interactions for a range of synthetic compounds with close attention to this specificity. It is important to note, however, that selectivity researchers observe in isolated in vitro systems does not guarantee equivalent behavior in complex in vivo environments.
Why Different Peptides Are Associated with Different Functions
Because distinct receptor subtypes appear in different tissue populations, and because peptide binding depends on the structural fit between a specific amino acid sequence and its receptor, individual peptides tend to align with distinct research areas in the scientific literature. As a result, the research community associates each peptide with a specific domain of inquiry rather than treating all peptides as interchangeable.
For example, researchers have examined growth hormone-releasing peptides in research exploring the hypothalamic-pituitary axis. Similarly, scientists have investigated antimicrobial peptides in the context of infectious disease and immune response research. Additionally, collagen-associated peptides have appeared in studies exploring extracellular matrix composition and integrity. In each case, the research association describes what researchers have studied, not what anyone has confirmed as an effective human application. Moreover, most of these investigations relied on in vitro models or animal subjects, and their findings cannot apply directly to human contexts without further controlled research.
What Peptide Therapy Is Commonly Promoted For
Across commercial websites, wellness platforms, and direct-to-consumer marketing channels, peptide therapy appears alongside a broad range of promoted applications. In practice, evaluating these promoted uses requires separating what the research literature has actually examined from what commercial language implies.
Repair, Recovery, and Tissue Research Discussions
Among the most frequently cited areas in peptide therapy discussions is the study of tissue repair and recovery processes. Certain compounds, particularly those researchers have investigated under the BPC (body protection compound) category, have appeared in peer-reviewed journals examining tissue-level responses in preclinical animal models. Specifically, these studies measured endpoints including wound site activity, connective tissue markers, and related biological signals under controlled laboratory conditions.
However, these findings arise almost entirely from animal-model research. No major regulatory authority has approved most of these compounds for any specific indication in humans. Furthermore, animal data does not confirm equivalent outcomes in human subjects. In addition, the gap between preclinical observations and verified human applications is significant, and commercial content that presents these findings as established outcomes for human use misrepresents the state of the evidence considerably.
Skin, Aging, Metabolism, and Performance-Related Claims
Commercial promotion of peptide therapy also routinely references skin structure, aging processes, metabolic function, and physical performance. Specifically, these areas draw selectively on fragments of the research literature without adequately representing the study population, the research model, or the preliminary nature of the underlying data.
For instance, research into collagen-related peptides has explored extracellular matrix composition in laboratory settings. Similarly, research into GLP-1 receptor agonist compounds has taken place in the context of metabolic regulation in investigational frameworks. In both cases, commercial marketing frequently extends findings from controlled research contexts into broad consumer-facing claims that the original studies do not support. As a result, researchers and informed readers should treat this gap as a standard feature of peptide marketing, not an exception.
Types of Peptides Often Mentioned in Peptide Therapy
Compound Category | Primary Research Area | General Regulatory Status |
|---|---|---|
Insulin and analogues | Metabolic regulation | Approved in multiple jurisdictions |
GLP-1 receptor agonists | Metabolic and appetite signaling | Some approved; others investigational |
Growth hormone-releasing peptides | Hypothalamic-pituitary axis | Generally unapproved for consumer use |
BPC-157 | Tissue repair, animal models | Not approved; research use only |
Antimicrobial peptides | Infectious disease, immune response | Varies by compound |
Collagen fragments | Extracellular matrix research | Not regulated as drugs in most markets |
Peptides Studied in Regulated Medical Contexts
Some peptides have received regulatory authorization for specific indications, and licensed physicians now use them in formal medical settings. Insulin represents the most globally recognized example. Vasopressin analogues, certain growth hormone-related compounds, and selected antimicrobial peptides have also undergone extensive study and, in some cases, regulatory bodies have authorized them for defined applications in specific jurisdictions.
By contrast, these authorized compounds differ fundamentally from the broader universe of research peptides. Specifically, their authorization reflects rigorous controlled evaluation, regulatory review of safety and efficacy data, and verified manufacturing standards. Importantly, the existence of authorized peptide pharmaceuticals does not imply that unapproved research compounds share equivalent evidence bases or safety records. In fact, conflating the two categories introduces significant analytical error that undermines accurate evaluation.
Research and Wellness Peptides Discussed in Commercial Settings
A substantial number of synthetic peptides circulate in commercial and wellness markets under research-use labeling. Compounds such as BPC-157, CJC-1295, Ipamorelin, and TB-500 are among those most frequently referenced. Researchers have examined most of them in in vitro systems or animal models, and a smaller number have entered early-phase investigations involving human subjects.
In fact, the legal status of these compounds varies meaningfully by country. In many jurisdictions, authorized use does not extend to personal consumer use, and suppliers may only sell them legally for laboratory research purposes. Their presence in wellness and consumer markets represents a regulatory gray zone that differs substantially from the frameworks governing authorized pharmaceutical compounds. Accordingly, buyers should understand this distinction clearly before making any sourcing decisions.
How Peptide Therapy Is Administered
Common Delivery Methods and Research Formats
In research and authorized medical contexts, researchers deliver peptides to biological targets through a range of routes that their biochemical properties determine. Subcutaneous and intravenous administration are common in research settings for compounds that gastrointestinal enzymes rapidly degrade when researchers deliver them orally. Some research teams have developed modified peptide sequences to improve oral stability, though achieving adequate bioavailability by this route remains a significant formulation challenge for many compound classes.
Researchers have also explored topical delivery for shorter peptide sequences, particularly in dermatology-related research involving skin structure and surface barrier function. Additionally, intranasal delivery has appeared in research contexts where central nervous system receptor access is relevant to the study design. All mention of delivery methods here is descriptive and appears strictly for research reference.
Why Administration Depends on the Peptide and Intended Use
In practice, the amino acid sequence, molecular weight, stability in biological fluids, and the receptor system that researchers target in the research model all determine the appropriate delivery route for any peptide. For instance, a compound that retains activity through one delivery format may show reduced activity through an alternative route because enzymatic activity at different anatomical sites degrades the compound before it reaches systemic circulation.
This variability is one key reason why generalizing across peptide compounds when discussing administration produces scientific inaccuracy. Consequently, each compound requires individual characterization based on its pharmacokinetic profile, as described in published research specific to that compound and the model in which researchers studied it.
Reported Effects and Where Claims May Be Overstated
Areas of Interest in Research and Scientific Practice
Published research into peptide compounds spans a broad and growing range of biological domains. Immune signaling, metabolic regulation, tissue modeling, central nervous system function, and antimicrobial activity have each generated substantial peer-reviewed literature examining peptide involvement. Notably, academic institutions, pharmaceutical research programs, and independent laboratories continue to produce new findings across these areas using increasingly sophisticated in vitro and in vivo models.
The scientific interest in peptides is real, measurable, and grounded in documented biological mechanisms. Indeed, the number of peer-reviewed publications on synthetic peptide compounds has increased substantially over the past two decades. Overall, this expanding literature deserves careful and accurate interpretation, with attention to model type, sample size, replication status, and the distance between preclinical observations and confirmed human outcomes.
Why Marketing Language Often Goes Beyond the Evidence
Overall, a consistent and documented pattern in commercial peptide markets involves the selective use of research literature to support product claims. Specifically, companies routinely present studies from cell cultures or rodent models as direct evidence of human outcomes without appropriate qualification. Moreover, they describe preliminary in vitro findings as established facts. In other words, research that identifies a mechanism in a controlled environment becomes, in commercial language, a promise of reliable and predictable results in general consumer contexts.
Indeed, regulatory agencies across multiple jurisdictions have noted this pattern. Furthermore, these agencies have issued enforcement actions and warning letters against companies making unsupported representations about unapproved peptide compounds in consumer-facing materials. As a result, anyone evaluating commercial peptide content should apply systematic scrutiny to any source presenting outcomes as certain, confirmed, or universally applicable.
Risks, Adverse Effects, and Important Limitations

Safety Concerns, Product Quality Issues, and Unknowns
The safety profile of most research peptides remains unestablished for human use. Specifically, animal model studies have identified various responses to peptide compounds depending on the specific compound, the species studied, the delivery route, and the duration of observation. In addition, long-term safety data in humans is largely absent for the majority of synthetic peptides currently circulating in commercial markets. Notably, the absence of reported adverse findings in short-term preclinical studies should not lead anyone to interpret this as a confirmation of human safety.
Product quality is a separate and serious concern. Independent analytical testing has documented cases where commercially marketed peptide compounds did not match their labeled purity or concentration. Contamination, sequence synthesis errors, and inaccurate labeling have all appeared through third-party verification processes. Furthermore, these quality issues compound the existing uncertainty around unapproved compounds and underscore the importance of sourcing from suppliers who provide verifiable analytical documentation.
Why Peptide Therapy Should Not Be Treated as a Universal Solution
Even where preclinical research results have shown noteworthy observations, no peptide compound can accurately characterize itself as a universal or broadly reliable intervention. Biological variability across research models, differences in delivery format, compound purity variation, and the fundamental gap between animal models and human physiology all introduce uncertainty that marketing language cannot resolve.
In practice, responsible engagement with peptide-related science requires acknowledging what researchers do not yet know alongside what they have observed. Distinguishing between a preliminary in vitro finding, a replicated animal study, and a confirmed human outcome is not a technicality. Rather, it is the foundational requirement for accurate scientific evaluation.
Legal, Regulatory, and Compliance Considerations
Regulated Use, Research Use, and Consumer Marketing Differences
In practice, peptide compounds occupy distinct legal categories depending on the jurisdiction and the intended use context. Specifically, some require physician authorization to use lawfully. Others have received authorization as pharmaceutical agents for specific, defined indications. Regulatory frameworks in many countries classify others as research chemicals, and suppliers may only sell them legally for laboratory research purposes, not for personal use by individuals.
Consumer marketing of unapproved peptides exists in a legally gray area that varies substantially by country. The FDA in the United States, the EMA in the European Union, and the TGA in Australia have each addressed the marketing of unapproved peptide compounds for human applications through guidance documents, warning letters, and enforcement actions. Therefore, researchers and buyers operating in this space should understand the regulatory landscape applicable to their jurisdiction before sourcing or distributing any compound.
Why Approval Status and Intended Use Matter
In essence, regulatory authorization indicates that a compound has completed controlled evaluation for safety and efficacy in defined populations under specific, reproducible conditions. By contrast, unapproved compounds have not completed this process regardless of how encouraging early-stage research results may appear or how prominently companies cite them in promotional materials.
Fundamentally, the distinction between research use and personal use carries meaningful legal and ethical weight. Research use implies controlled laboratory conditions, qualified personnel, appropriate institutional oversight, and compliance with applicable research regulations. Blurring this distinction introduces legal risk and, moreover, undermines the integrity of the research framework that gives peptide science its credibility.
Peptide Therapy and Peptide Researches

How Peptide Researches Supports Research-Focused Sourcing, Documentation, and Compliance-First Communication
Specifically, Peptide Researches operates as a supplier of peptide compounds for laboratory and scientific research purposes. The platform structures every aspect of its sourcing, documentation, and communication around transparency and compliance with applicable standards for research-grade materials.
Peptide Researches accompanies all listed compounds with certificates of analysis (COAs) that include purity data, molecular weight confirmation, and sequence verification. As a result, this analytical documentation allows researchers to evaluate compound quality as part of their standard laboratory procurement process. Furthermore, researchers who require documented, verifiable material for in vitro or in vivo studies can access this information directly through the platform.
Importantly, Peptide Researches does not make health claims, personal-use representations, or outcome promises of any kind. Accordingly, the company writes all content for research and laboratory audiences, and sells all materials exclusively for research use. This approach reflects a considered commitment to responsible communication in a space where misleading and non-compliant marketing has become widespread.
Conclusion
Peptide therapy is a term that spans a wide range of scientific, medical, and commercial contexts, and its meaning shifts significantly across each. Distinguishing between authorized pharmaceutical applications, preclinical research findings, and consumer-facing commercial claims is not optional for anyone seeking to engage with this area of science accurately.
The research literature on peptides is real, growing, and grounded in reproducible molecular biology. However, the distance between what researchers have observed in controlled laboratory models and what anyone can confirm as a reliable human application remains substantial for most compounds that companies discuss commercially under the peptide therapy label.
Therefore, researchers and procurement professionals evaluating peptide-related content should rely on peer-reviewed literature, verified analytical documentation, and regulatory guidance as their primary reference points. Peptide Researches provides research-grade compounds with full analytical documentation to support rigorous, compliant laboratory research across peptide-related scientific disciplines.