Researchers, regulators, and commercial marketers each apply the phrase "peptides for weight loss" in very different ways and for very different purposes. In regulated research settings, the phrase points to specific compound classes that scientists have studied for their influence on appetite signaling, gastric motility, and metabolic regulation. In commercial contexts, however, the same phrase appears alongside claims that the available evidence frequently does not support.
This article explains what peptides researchers associate with body weight regulation, how these compounds are described to function at the biological level, what the published evidence currently says, and what legal and sourcing considerations matter most. All content here is produced for educational and laboratory research reference purposes only.
What People Mean by Peptides for Weight Loss
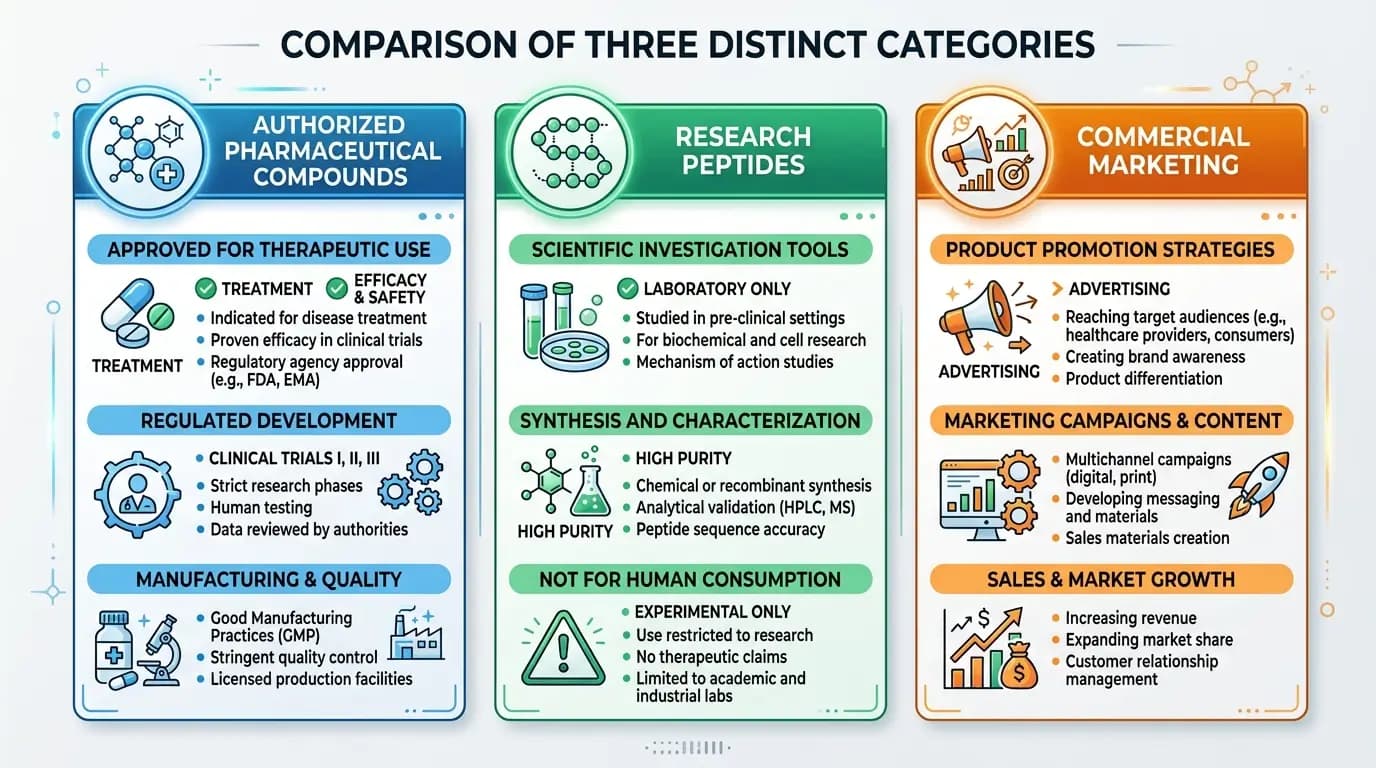
The phrase "peptides for weight loss" covers at least three distinct categories of compounds that researchers, clinicians, and marketers discuss in very different frameworks. Specifically, these categories include authorized pharmaceutical compounds, research-grade peptides studied in preclinical or investigational contexts, and commercially promoted products whose claims often move ahead of any credible evidence base.
Why Weight Loss Peptides Are Usually Linked to Appetite and Metabolic Pathways
Researchers link specific peptide compounds to body weight regulation primarily because of how these compounds interact with biological systems that govern hunger, satiety, and energy metabolism. Several naturally occurring peptides in the human body, including glucagon-like peptide 1 (GLP-1), peptide YY (PYY), and ghrelin, play documented roles in appetite signaling. As a result, synthetic compounds that interact with the receptors for these peptides have become subjects of active investigation in the context of body composition research.
GLP-1, in particular, represents one of the most well-studied appetite-related peptide pathways. Notably, the small intestine produces this peptide in response to nutrient intake, and its receptor engagement slows gastric emptying, stimulates insulin secretion, and reduces appetite signaling in the central nervous system. These mechanisms, individually and in combination, have attracted significant research interest in the context of body weight regulation.
The Difference Between Authorized Peptides, Research Peptides, and Marketing Language
Understanding the difference between these three categories is, fundamentally, essential for evaluating any claim made about peptides in the context of body weight research. Authorized pharmaceutical compounds are those that regulatory agencies have reviewed and approved for specific indications in human populations. Research peptides are compounds that researchers study in laboratory or investigational settings, typically in in vitro systems or animal models, and these have not completed full regulatory review for human applications. Commercial marketing language, by contrast, applies peptide-related terminology to products and services regardless of whether the underlying evidence supports any specific outcome.
Conflating these three categories introduces significant misunderstanding. Accordingly, anyone evaluating content about peptides and body weight regulation should identify which category the specific compound falls into before drawing any conclusion about what the evidence supports.
How Peptides for Weight Loss Are Described to Work
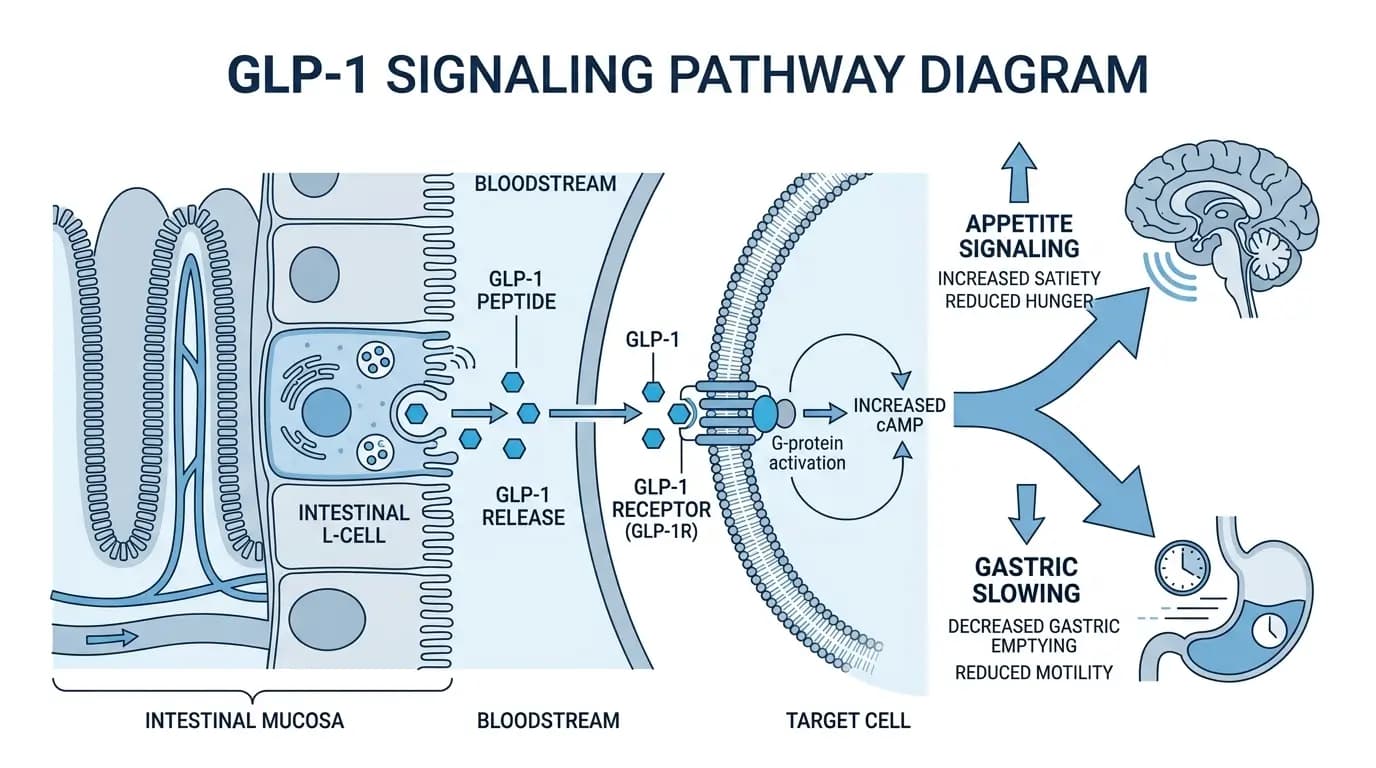
Researchers describe the compounds most frequently discussed in body weight regulation research as functioning through receptor-mediated biological pathways that influence appetite, energy intake, and metabolic signaling. The plausibility of these descriptions varies considerably depending on the specific compound and the research model in which researchers have studied it.
Appetite Regulation, Gastric Emptying, and Blood Sugar Effects
The most well-characterized mechanism among body weight-related peptide compounds involves GLP-1 receptor engagement. When synthetic GLP-1 receptor agonist compounds bind to GLP-1 receptors, they produce a constellation of documented effects in controlled research settings. Specifically, these include delayed gastric emptying, which prolongs the physical sensation of fullness after nutrient intake; enhanced insulin secretion in response to elevated blood glucose; and reduced appetite signaling through central nervous system pathways.
In controlled research models, these combined effects translate into reduced caloric intake over time. Furthermore, researchers have noted that compounds with longer half-lives produce more sustained receptor engagement, which extends the duration of appetite suppression observed in study participants. These mechanisms form the scientific basis for the most credible research into body composition and peptide interactions published in peer-reviewed literature to date.
Other peptide systems researchers discuss in this context include peptide YY (PYY), which the intestinal L-cells release after nutrient intake and which suppresses appetite through hypothalamic pathways. Additionally, researchers have examined ghrelin-antagonist approaches, since ghrelin is the primary appetite-stimulating peptide and reducing its activity represents an alternative mechanistic strategy in body composition research.
Why Some Peptides Are Discussed for Body Fat Research While Others Are Not
Not all peptides attract research interest in the context of body weight regulation. The compounds that researchers focus on most consistently are those with documented interactions with receptor systems that play established roles in energy homeostasis. By contrast, peptides that act on receptor systems unrelated to appetite, energy metabolism, or adiposity regulation do not have a plausible mechanistic basis for discussion in body composition research.
In practice, commercial marketing frequently extends peptide-related terminology into domains where the mechanistic rationale is weak or absent. For example, some growth hormone-releasing peptides attract commercial discussion in body composition contexts despite having mechanistic pathways that differ significantly from those of GLP-1-related compounds. In each case, researchers should evaluate the specific receptor system involved, the research model used, and the quality of the available literature before drawing conclusions.
Common Peptides Mentioned in Weight Loss Conversations
Compound or Class | Receptor System | Research Setting | General Regulatory Status |
|---|---|---|---|
GLP-1 receptor agonists | GLP-1 receptor | Controlled trials, some authorized use | Some authorized; others investigational |
Liraglutide | GLP-1 receptor | Controlled investigational trials | Authorized in multiple jurisdictions |
Peptide YY analogues | Y2 receptor | Preclinical and early investigational | Largely investigational |
Ghrelin antagonists | Ghrelin receptor | Primarily preclinical | Not authorized |
Growth hormone-releasing peptides | GHRH receptor | Primarily preclinical | Not authorized for consumer use |
GLP-1-Related Peptide Classes and Key Research Compounds
GLP-1 receptor agonist compounds represent the category with the strongest and most consistent evidence base in body weight regulation research. Liraglutide, for instance, has undergone controlled investigational research in body weight regulation contexts, and regulatory agencies in multiple jurisdictions have authorized specific formulations for defined indications in adult populations. The evidence supporting liraglutide in body composition research contexts comes from randomized controlled trials published in major peer-reviewed journals, including the NEJM.
Importantly, the fact that some GLP-1-related compounds have received regulatory authorization does not mean that all compounds marketed under GLP-1-related language carry equivalent evidence. Research into longer-acting GLP-1 receptor agonist compounds and dual agonist peptides continues across multiple investigational frameworks. However, the research status of investigational compounds differs fundamentally from the authorized status of approved pharmaceutical agents, and commercial marketing frequently blurs this distinction in ways that mislead both researchers and consumers.
Researchers studying GLP-1 receptor agonist compounds in controlled laboratory settings can review the Sema peptide listing at Peptide Researches for verified analytical documentation, sequence confirmation, and research-use sourcing information.
Newer Compounds and Why Some Claims Move Ahead of the Evidence
Beyond GLP-1 receptor agonists, researchers have examined a range of newer peptide compounds and dual-receptor agonist structures in body weight regulation contexts. Notably, compounds designed to engage GIP, GLP-1, and glucagon receptor pathways simultaneously represent one of the more actively studied multi-receptor agonist classes in recent preclinical and early-phase investigational research. Researchers examining this compound class in controlled laboratory settings can review the multi-receptor agonist research peptide listing at Peptide Researches for analytical documentation and research-use sourcing details. Some of these investigational compounds have shown notable findings in early-phase or animal-model research. As a result, commercial interest in these compounds has grown substantially, often ahead of the evidence base that would justify any specific claim about human outcomes.
Indeed, a recurring pattern in this area involves commercial marketing that references early-phase research findings as confirmation of efficacy, when in fact these findings remain preliminary and subject to replication in larger, more rigorous research frameworks. Furthermore, several investigational compounds in this category have entered commercial markets under research-use labeling without completing the controlled research process that regulatory authorization requires. Researchers and informed buyers should, therefore, treat claims about newer compounds with particular caution until peer-reviewed evidence from appropriately designed studies becomes available.
What the Current Evidence Says About Peptides for Weight Loss
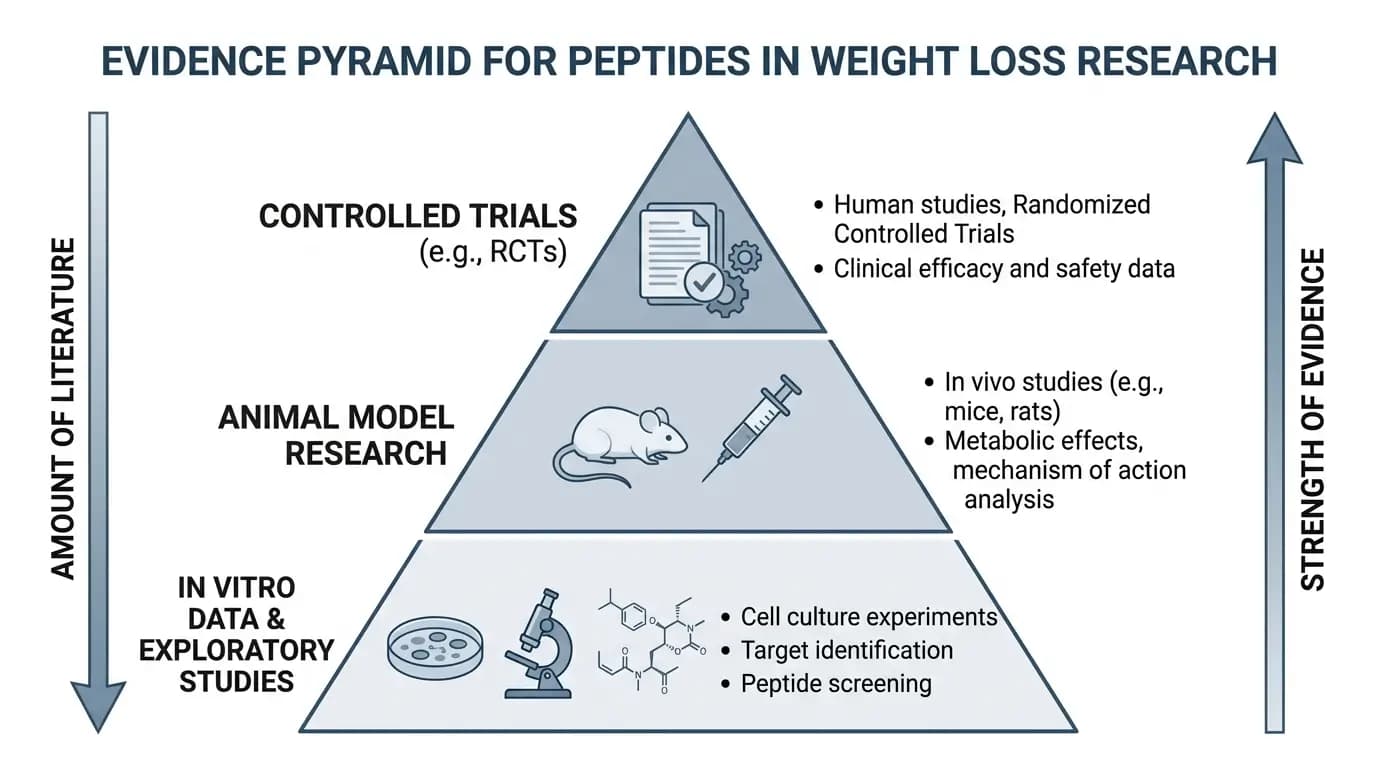
Where the Strongest Evidence Exists Today
The strongest and most consistently replicated evidence in the intersection of peptide research and body weight regulation comes from GLP-1 receptor agonist research. Randomized controlled trials, systematic reviews, and regulatory submissions have established a credible evidence base for specific GLP-1-related compounds in defined adult populations meeting specific research eligibility criteria. Notably, this evidence comes from controlled settings with structured monitoring, defined endpoints, and documented follow-up periods, not from general consumer use.
In practice, the research literature also supports the biological plausibility of several mechanistic pathways connecting peptide receptor engagement to metabolic regulation and body weight outcomes. Specifically, the gastric emptying effects, appetite signaling modulation, and insulin secretion responses documented in GLP-1 research have all been replicated across multiple independent research programs. This makes GLP-1-related compounds the most credible reference point when evaluating any discussion of peptides and body composition research.
Why Not Every Peptide Promoted for Body Composition Has the Same Level of Support
By contrast, many peptides that commercial channels discuss in body composition contexts do not have equivalent evidence. Growth hormone-releasing peptides, for instance, have generated research interest in areas including body composition, but their evidence base derives primarily from preclinical models rather than the kind of controlled human research that regulatory review requires. Similarly, peptide YY analogues remain largely investigational, with promising but limited preliminary data.
Moreover, some commercial products claim body composition effects without credible published research of any kind. In these cases, claims rest on mechanistic speculation, anecdote, or misrepresented data from studies conducted under very different conditions. Overall, researchers evaluating the evidence landscape should demand clear identification of the research model, the study population, and the publication source before treating any claim as substantiated.
Risks, Adverse Effects, and Major Limitations
Common Adverse Effects and Tolerability Concerns
The authorized GLP-1 receptor agonist compounds that regulatory agencies have reviewed have documented adverse effect profiles established through controlled research. Gastrointestinal responses, including nausea, reduced appetite, and changes in gastric motility, represent the most frequently documented adverse effects in published research. Furthermore, the controlled research literature also documents tolerability patterns across different graduated escalation schedules and in different adult population subgroups.
For research-grade peptides that have not completed regulatory review, however, researchers face a different situation. In many cases, the adverse effect profile for these compounds in human settings remains largely undocumented. Short-term preclinical findings do not reliably predict human adverse effect profiles. Accordingly, sourcing and using unapproved research compounds outside controlled laboratory settings introduces significant and unquantified uncertainty.
Product quality adds another layer of risk. Independent analytical testing has documented cases where commercially marketed peptide compounds did not match their labeled purity or sequence. Specifically, contamination, synthesis errors, and concentration discrepancies have all appeared in third-party verification programs. These quality issues compound the existing uncertainty around unapproved compounds considerably.
Why Eligibility Assessment, Monitoring, and Long-Term Expectations Matter
Research into body weight regulation with peptide compounds consistently demonstrates that outcomes vary significantly across different research populations. Specifically, the controlled trials that established the most credible findings in this area enrolled adult populations meeting defined eligibility criteria, with structured monitoring, graduated adjustments, and follow-up protocols. In other words, the evidence does not support generalizing controlled research findings to unrestricted general use.
Long-term expectations also require careful management. In controlled research, body weight regulation effects appear to persist only while compound administration continues. Furthermore, withdrawal from GLP-1-related compounds in controlled research contexts has associated with gradual return toward baseline body weight measurements. These findings underscore the importance of framing body composition research outcomes accurately rather than through the lens of permanent change.
Legal, Regulatory, and Compliance Considerations
Why Some Weight Loss Peptides Require Medical Supervision
Regulatory frameworks in multiple jurisdictions require that GLP-1 receptor agonist compounds authorized for body weight regulation applications be used under licensed physician oversight. These requirements exist because authorized compounds in this category carry documented adverse effect profiles, require structured monitoring, and involve eligibility criteria that require medical evaluation. Therefore, access to authorized compounds in this class requires physician authorization in most countries where they are approved.
Obtaining these compounds through unregulated channels, including online markets that do not require physician oversight, introduces significant legal and safety risk. The FDA in the United States, the EMA in the European Union, and comparable agencies elsewhere have each issued guidance and enforcement actions addressing unauthorized access to and marketing of body weight regulation compounds.
Why Research-Use Products Should Not Be Confused With Authorized Pharmaceutical Compounds
Research-grade peptides sold for laboratory use occupy a fundamentally different legal and scientific category from authorized pharmaceutical compounds. Specifically, research-grade compounds have not completed regulatory review for human applications, do not carry authorized indications, and are not intended or labeled for personal use. Suppliers legally sell these compounds only for controlled laboratory research purposes.
Commercial marketing that uses authorized compound names or regulatory terminology to describe research-grade products misrepresents both the compounds and their legal status. Indeed, several regulatory agencies have explicitly addressed this misrepresentation in enforcement actions targeting companies that market research compounds using language implying equivalence to authorized pharmaceutical agents. Researchers and informed buyers should, accordingly, understand this distinction clearly before engaging with any supplier or product in this space.
How to Evaluate Claims About Peptides for Weight Loss
Red Flags in Fast Body-Change Promises and Before-and-After Marketing
The body composition research and marketing space contains a high volume of misleading content. Specifically, certain patterns consistently indicate that a source is misrepresenting the evidence. Claims that a compound produces rapid body composition changes without physician oversight, qualification criteria, or structured monitoring contradict what controlled research actually demonstrates. Similarly, before-and-after marketing relies on individual outcomes that cannot characterize population-level research findings.
Furthermore, any source that presents preliminary or preclinical research findings as confirmation of human efficacy is misrepresenting the evidence. In addition, sources that use authorized compound names alongside unapproved research products to imply equivalence are engaging in a form of misrepresentation that regulators have specifically addressed. Overall, the absence of peer-reviewed citations, independent verification, and transparent regulatory status disclosure are all strong signals that a source lacks credibility.
What Transparency, Labeling, and Evidence Signals Matter Most
Credible sources in this research area share several characteristics. Specifically, they identify the research model behind any referenced finding clearly, distinguish authorized pharmaceutical applications from research-use contexts, and present evidence from peer-reviewed publications with verifiable citations. Moreover, credible suppliers provide certificates of analysis, independent purity verification, and research-use-only labeling that accurately reflects the regulatory status of the compounds they provide.
In practice, the most reliable signal of a credible source is its willingness to present limitations alongside findings. Any source that presents peptide research as free of uncertainty, limitation, or regulatory complexity is not engaging honestly with the published literature. By contrast, sources that acknowledge what research has and has not established, and that present evidence in proportion to its actual strength, provide a significantly more reliable foundation for research-informed decision-making.
Peptides for Weight Loss and Peptide Researches
How Peptide Researches Supports Research-Focused Sourcing, Documentation, and Compliance-First Communication
Peptide Researches provides research-grade peptide compounds for laboratory and scientific research purposes. Specifically, the platform structures its sourcing, documentation, and communication around full transparency, compliance with applicable research-grade standards, and accurate representation of compound regulatory status. All compounds listed on the platform come with certificates of analysis (COAs) that include purity data, molecular weight confirmation, and sequence verification.
Furthermore, Peptide Researches does not make body composition claims, outcome representations, or personal-use guidance of any kind. All content on the platform addresses research audiences, and all materials are sold exclusively for laboratory research use. This approach reflects a commitment to responsible sourcing communication in a market where misleading claims and non-compliant product representations have become widespread. Researchers who require analytically documented, research-grade compounds for in vitro or in vivo study can access verifiable documentation directly through the platform.
Among the research-grade compounds available at Peptide Researches relevant to body weight regulation research, the Sema peptide and the multi-receptor agonist research peptide each include full certificates of analysis, purity data, and sequence verification for laboratory research use only.
Conclusion
The phrase "peptides for weight loss" spans a wide range of scientific, regulatory, and commercial contexts, and its accuracy depends entirely on which compound, which research model, and which regulatory framework researchers are discussing. Specifically, the distinction between authorized pharmaceutical compounds with established research records, investigational compounds with preliminary findings, and commercially promoted products with no credible evidence base is not optional for anyone engaging with this topic accurately.
Furthermore, the published research in this area is real, growing, and grounded in documented biological mechanisms. However, the evidence base varies enormously across different compounds, and responsible engagement requires applying that variability accurately rather than treating all "weight loss peptides" as a single category with uniform support.
Therefore, researchers and procurement professionals evaluating this space should rely on peer-reviewed literature, regulatory documentation, and verified analytical records as their primary reference points. Peptide Researches provides research-grade compounds with full analytical documentation to support rigorous, compliant laboratory research across peptide-related scientific disciplines.
References
Drucker DJ. The biology of incretin hormones. Cell Metab. 2006;3(3):153-165.
Baggio LL, Drucker DJ. Biology of incretins: GLP-1 and GIP. Gastroenterology. 2007;132(6):2131-2157.
Müller TD, Finan B, Bloom SR, et al. Glucagon-like peptide 1 (GLP-1). Mol Metab. 2019;30:72-130.
Holst JJ. The physiology of glucagon-like peptide 1. Physiol Rev. 2007;87(4):1409-1439.