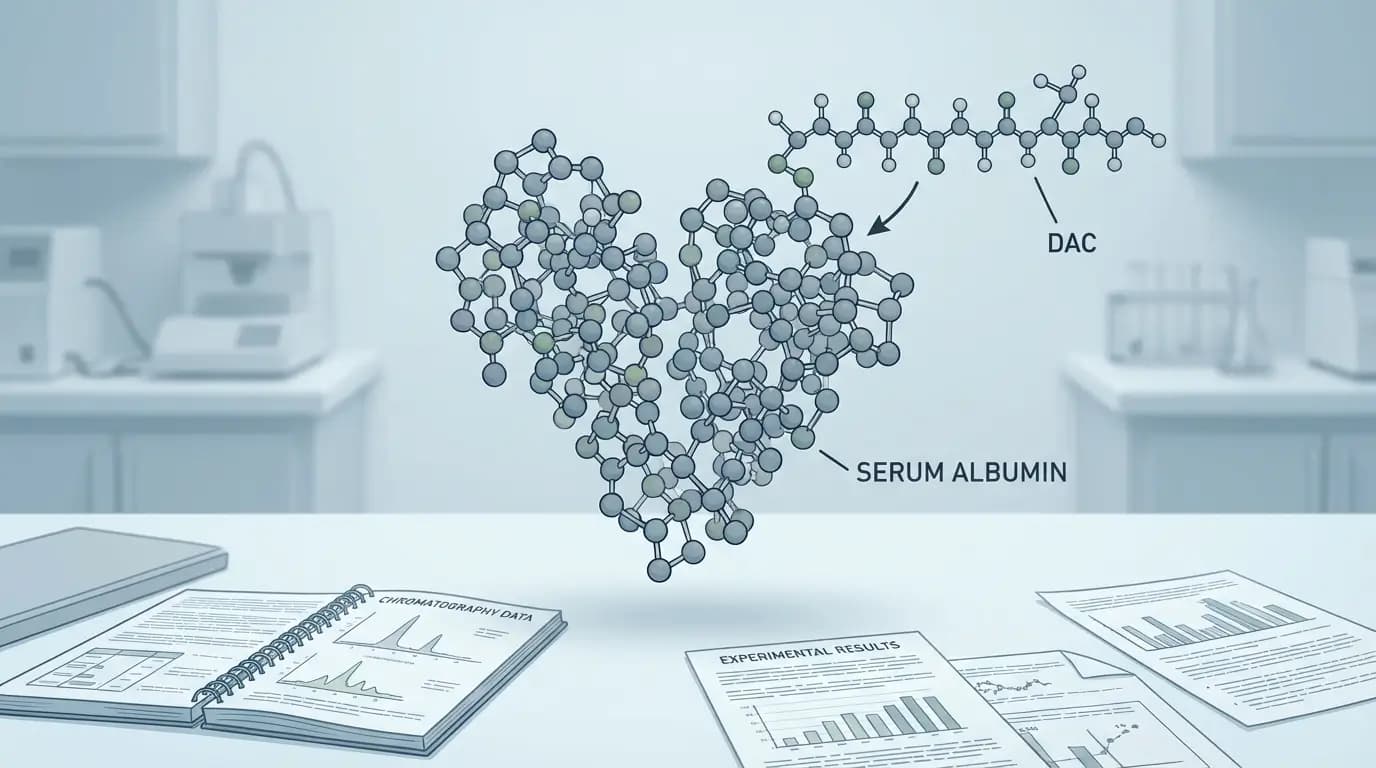
DAC in peptides refers to a chemical modification designed to change how a peptide behaves in circulation. When researchers ask what is DAC in peptides, they usually mean a Drug Affinity Complex, a structure built to help a peptide attach to serum albumin and remain present longer than an unmodified sequence.
Introduction
The question what is DAC in peptides appears often because many catalog pages mention DAC without explaining the chemistry behind the label. In simple terms, DAC is not a separate peptide family. Instead, this modification attaches to a peptide, enabling the final construct to react with albumin, a major blood protein that circulates longer than many short peptides.
Because the body clears many peptides quickly, researchers have long explored ways to slow degradation and reduce rapid kidney loss. Researchers developed DAC as one of those strategies. ConjuChem describes DAC technology as a way to address peptidase degradation and fast excretion, and the early DAC literature shows this idea in practice with albumin-linked GLP-1 and growth hormone-releasing factor analogs.
What Is DAC in Peptides, in Simple Terms?

What DAC stands for
DAC stands for Drug Affinity Complex. In other words, the name describes a peptide construct that keeps its active peptide portion while gaining a reactive chemical group and linker that allow binding to albumin. ConjuChem breaks the construct into three parts: the active drug component, a linker, and a reactive chemistry group, often a maleimide selected for thiol reactivity.
When people search what is DAC in peptides, they sometimes assume DAC is a synonym for CJC-1295. That is not correct. CJC-1295 is one peptide analog that uses DAC, while older DAC publications also describe CJC-1131, a GLP-1 analog designed to conjugate to albumin in vivo.
Why DAC is a modification, not a standalone peptide
Its amino acid sequence usually defines a standalone peptide first. DAC changes that picture. The peptide sequence remains central, yet the finished construct also includes a linker and a reactive end group that extend beyond the base sequence itself. Therefore, when a catalog says "with DAC," it describes a modified version of a peptide, not an unrelated molecule with a similar name.
This point matters for labeling, literature reading, and comparison work. A peptide listed with DAC and the same peptide listed without DAC may share a core sequence or analog family, but they are not chemically identical, and their circulation profile, albumin interaction, and study design logic may differ in important ways.
Why Researchers Use DAC in Peptide Design
The short-life problem in many peptides
Many peptides are small, flexible molecules that face two common challenges in circulation: enzymatic breakdown and fast clearance. ConjuChem's description of DAC technology focuses on those same barriers, namely peptidase degradation and rapid kidney excretion. Likewise, early GLP-1 DAC papers centered on the idea that native GLP-1 degrades rapidly, pushing researchers toward more stable analogs and albumin-linked constructs.
When someone asks what is DAC in peptides, the practical answer often starts here. DAC is one strategy for keeping a peptide in circulation longer without turning it into a full protein fusion. Because albumin already circulates widely and persists far longer than many peptide fragments, albumin attachment offers a way to change peptide pharmacokinetics through carrier binding rather than by completely redesigning the peptide backbone.
Why albumin matters
Albumin is attractive in peptide engineering because it is abundant in plasma and has a reactive cysteine residue, Cys34, that can serve as an anchor point for appropriately designed chemistry. ConjuChem states that DAC constructs react selectively with albumin and highlights the free thiol on cysteine-34 as the main attachment site. The original CJC-1295 and CJC-1131 papers follow that same concept and describe albumin bioconjugation as the route to longer plasma presence.
How DAC Changes Peptide Structure
The three-part DAC construct
A useful way to understand what is DAC in peptides is to picture three connected pieces. First comes the peptide portion that carries the intended receptor activity in the published model. Second comes a linker. Third comes a reactive group, usually a maleimide or related thiol-reactive chemistry, placed at the far end of the linker. ConjuChem presents exactly that three-part structure in its DAC overview.
The early CJC-1295 paper gives a concrete example. The authors describe CJC-1295 as a tetrasubstituted hGRF(1-29) analog with an added N epsilon-3-maleimidopropionamide derivative of lysine at the C terminus. In parallel, the CJC-1131 GLP-1 papers describe maleimide-bearing lysine derivatives added so the peptide can bioconjugate to albumin at Cys34.
Why the C-terminus shows up so often
The C-terminus appears often in DAC examples because that region can host an extra lysine or linker arrangement without erasing the intended receptor-recognition region described in the paper. In the CJC-1131 work, the point of albumin attachment at the C-terminal end gave the best combination of stability and bioactivity among the tested analogs. Similarly, the CJC-1295 paper places the reactive addition at the C terminus of the hGRF analog.
How DAC Works After Albumin Binding

Binding to albumin
Once the DAC-bearing peptide encounters albumin, the reactive chemistry can form a covalent link with the free thiol at albumin Cys34. ConjuChem states that the attachment is permanent and does not require the peptide to detach from albumin to retain biological function. The original CJC-1295 paper also detected an immunoreactive species corresponding to serum albumin in rat plasma shortly after administration, which supports in vivo albumin conjugation for that analog.
Slower clearance and greater stability
Published DAC papers repeatedly connect albumin binding with slower clearance and greater stability. ConjuChem states that albumin conjugation can block cleavage by peptidases and multiple clearance pathways. The CJC-1295 rat paper reported plasma presence beyond 72 hours, while the 2006 adult-volunteer study estimated a half-life of 5.8 to 8.1 days for CJC-1295. A later paper discussing adult subjects described an 8 to 10 day half-life and linked that duration to albumin binding.
That said, longer circulation does not automatically tell a researcher everything important about a peptide. Receptor selectivity, tissue distribution, assay design, and the specific model still matter. Therefore, when readers ask what DAC is in peptides, the accurate answer is not only "a half-life extender," but a defined albumin-binding modification that changes pharmacokinetics and requires interpretation within the full experimental context.
CJC-1295 as the Best-Known DAC Example
Why CJC-1295 dominates this topic
CJC-1295 dominates the question of what DAC is in peptides because published research widely cites the peptide and commercial catalogs widely label it. In the 2005 Endocrinology paper, researchers identified CJC-1295 as a stable and active hGRF(1-29) analog with an extended plasma half-life. In the 2006 JCEM paper, healthy adult volunteers showed sustained GH and IGF-I changes after CJC-1295 administration, and the paper reported an estimated half-life in the multi-day range.
Many readers first encounter DAC through product names such as "CJC-1295 with DAC." However, the published literature makes clear that DAC is the albumin-binding design element, while CJC-1295 is the peptide analog carrying that element.
What the papers actually showed
The early CJC-1295 paper used cultured rat pituitary cells and normal male Sprague Dawley rats. The authors reported enhanced in vitro stability against DPP-IV, measurable GH secretion in rats, and plasma presence beyond 72 hours for the selected analog. The later adult-volunteer paper reported sustained GH and IGF-I changes after single administrations, with an estimated half-life of 5.8 to 8.1 days.
These findings are important, but the limits matter just as much. The earlier work combined cell assays and animal models. The later work involved healthy adults in a published study, not every possible population, and it focused on measured hormone markers over defined observation periods. In short, the papers explain why DAC changed the research conversation around CJC-1295, yet they do not justify broad claims across unrelated peptides.
CJC-1295 With DAC vs Without DAC

The basic difference
This is where many readers revisit the question what is DAC in peptides. CJC-1295 with DAC contains the added linker and reactive chemistry intended for albumin conjugation. A version without DAC lacks that albumin-binding addition, so it should not be expected to share the same persistence in circulation. The published CJC-1295 literature explains the longer half-life through albumin binding, which is the defining difference between the DAC-labeled form and a shorter analog without that feature.
A simple comparison table
The table below is a simplified reading aid for catalogs and research notes. It summarizes how the two labels are usually interpreted in the literature and in catalog naming, rather than giving instructions for use.
Feature | CJC-1295 with DAC | CJC-1295 without DAC |
|---|---|---|
Structural idea | Core analog plus albumin-binding DAC chemistry | Core analog without albumin-binding DAC chemistry |
Albumin interaction | Designed for albumin conjugation | No DAC-driven albumin conjugation |
Expected circulation profile | Longer persistence in published DAC papers | Shorter persistence expected than DAC form |
Why catalogs separate them | Modification changes identity and kinetics | Base analog is discussed separately |
This distinction matters because a name difference on a vial label may reflect a real chemistry difference, not a trivial branding choice. Likewise, any comparison should start with the sequence, terminal modifications, and literature source rather than with assumptions based on shorthand product names.
Why the comparison can confuse beginners
Beginners often see "with DAC" and think the phrase refers to greater potency. The literature does not frame DAC that way. Instead, it frames DAC as a strategy for albumin conjugation, longer plasma presence, and altered pharmacokinetics. Therefore, the cleaner beginner answer to what is DAC in peptides is this: DAC changes how long the peptide stays around, not simply what the peptide is called.
What Published Studies Say About DAC-Modified Peptides
DAC beyond CJC-1295
CJC-1295 is the best-known DAC example, yet it is not the only one. Before the CJC-1295 work, DAC had already appeared in GLP-1 analog research through CJC-1131. In the 2003 Diabetes paper, the authors described CJC-1131 as a DPP-IV-resistant DAC GLP-1 compound that conjugates to albumin in vivo and showed receptor activation in cell systems as well as activity in mouse models. A 2004 follow-up paper identified the C-terminally attached version as the best balance of stability and bioactivity among the analogs they tested.
What the literature supports, and what it does not
The literature supports several narrow points with reasonable clarity. DAC refers to a peptide construct with a linker and reactive group. The construct is designed to bind albumin, often through Cys34. Published DAC examples show longer plasma presence than short unmodified peptide forms. These are strong, source-backed takeaways.
However, the literature does not support careless generalization. It does not show that every peptide "needs" DAC. It does not show that any peptide becomes interchangeable with another once albumin binding is added. And it certainly does not support turning a chemistry discussion into a consumer-outcome promise. In research writing, the strongest approach is to stay close to the specific sequence, the exact modification, and the exact model in the cited paper.
How to Read DAC on a Product Page or COA

Start with the identity details
When a product page says DAC, the first task is to verify identity details rather than stopping at the headline. For example, check whether the sequence is listed, whether a molecular formula or molecular weight is provided, and whether the modification site is described clearly at the terminus or linker level. Because DAC adds real chemistry to the parent peptide, vague naming alone is not enough for rigorous catalog reading.
A careful reader will usually check these points:
Peptide name and full variant name
Sequence or analog description
Modification site, linker, or reactive group details
Molecular weight and purity listing
COA availability and batch-specific analytics
Storage wording and handling wording written for laboratory context
These details help separate a well-described DAC construct from a vague label that leaves too much unstated. Moreover, they reduce confusion when two similarly named peptides differ only by terminal chemistry.
Why COA reading still matters
The phrase what is DAC in peptides often leads readers straight to mechanism. A COA will not prove every literature claim, yet it can confirm whether the material matches the named construct, whether purity is stated, and whether analytical testing is documented for that batch. Therefore, DAC should be read as part of a broader specification set, not as a standalone quality signal.
Common Misunderstandings About DAC
DAC does not mean every peptide behaves the same way
One of the biggest mistakes around what is DAC in peptides is assuming DAC creates a universal behavior across all sequences. It does not. Albumin binding may be the shared idea, but the peptide family, receptor biology, modification site, assay method, and study model still determine the final behavior. The GLP-1 DAC papers and the CJC-1295 DAC papers show that the same platform logic can appear in different peptide contexts, yet they are still distinct research stories.
DAC is not a shortcut around experimental design
Another misunderstanding is that longer plasma presence makes design choices easy. Actually, it can make them more important. Once kinetics change, assay timing, sampling windows, and interpretation windows also change. Therefore, asking what is DAC in peptides should lead to a second question: how does this modification change the meaning of the readout in this specific experiment? That is a research-method question, not a branding question.
DAC should be discussed with limits in view
The most reliable DAC writing keeps the limits visible. Some papers involve cultured cells, some involve rodents, and some involve healthy adults under defined study conditions. Those settings are informative, but they are not interchangeable. In conclusion, the strongest DAC explanation stays close to the source material and avoids sweeping claims.
Conclusion
What is DAC in peptides? The simplest answer is that DAC is a Drug Affinity Complex, a chemical modification added to a peptide so it can conjugate to albumin and remain in circulation longer than an unmodified form. The published DAC literature, especially around CJC-1131 and CJC-1295, consistently ties DAC to albumin binding, slower clearance, and altered pharmacokinetics rather than to a completely separate peptide identity.
For beginners, the key takeaway is to read DAC as chemistry, not as hype. For researchers, the next step is to check the exact analog, the terminal modification, the model used in the cited paper, and the batch-specific analytical documents attached to the material. Overall, that approach gives the cleanest answer to what is DAC in peptides and helps avoid confusion between a base peptide and its albumin-binding variant.