For laboratory research use only. Not intended for use in humans or animals. Not intended to diagnose, treat, cure, or prevent any disease.
Retatrutide is an investigational peptide researchers study for its unusual three-target design. Current literature describes it as a single molecule that activates the GIP receptor, the GLP-1 receptor, and the glucagon receptor, which is why many papers call it a triple agonist or tri-agonist. In simple terms, the topic matters because most earlier compounds in this area were built around one or two pathways, while retatrutide was designed to engage three at once.
Search interest has grown because early published data drew attention to the scale of change seen in measured study endpoints over 24 and 48 weeks in a Phase 2 trial. At the same time, late-stage studies are still ongoing, so the evidence base is still developing and should be read as a research story in progress rather than a finished regulatory story.
What retatrutide is in research terms
A synthetic triple-agonist peptide
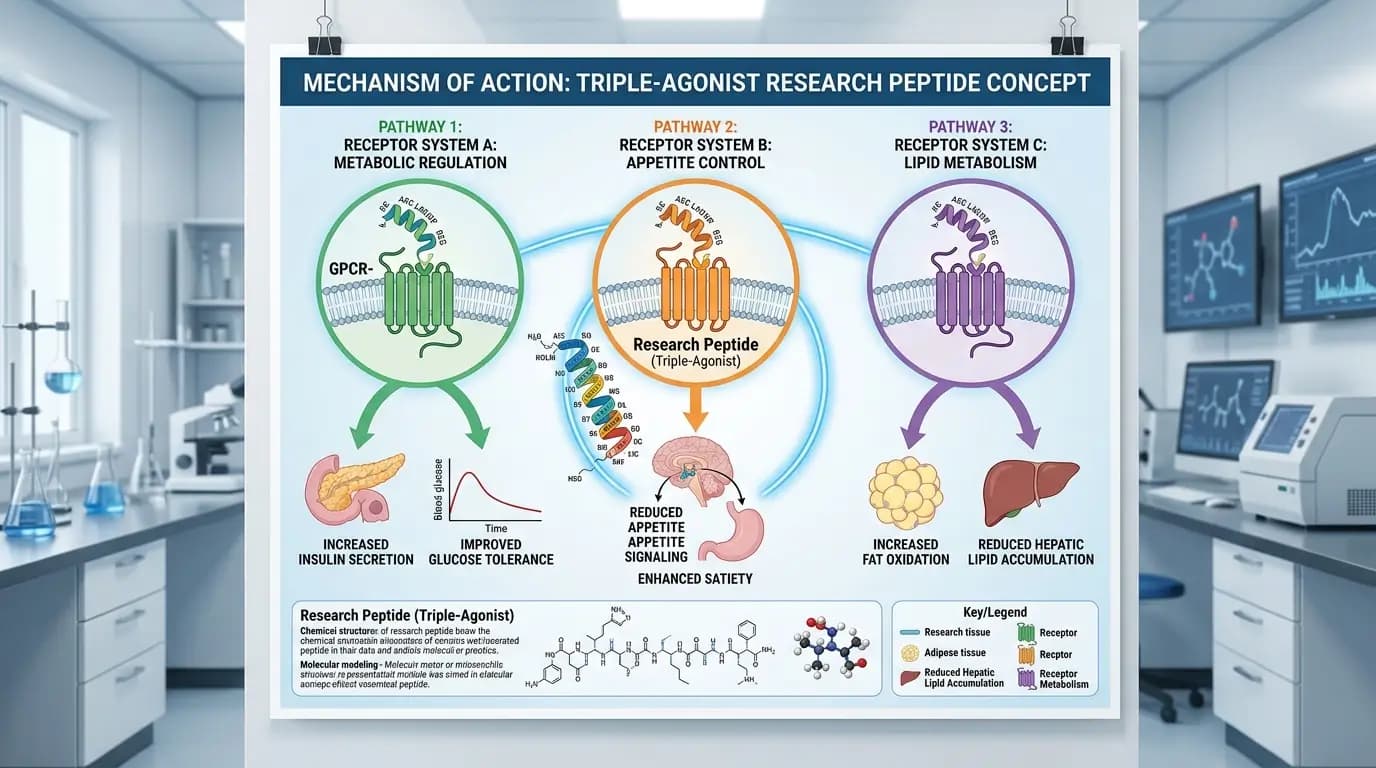
In the literature, retatrutide is described as a synthetic peptide engineered to activate three receptor systems tied to metabolic signaling. Those receptors are GIP, GLP-1, and glucagon. That three-part design is the central reason the molecule stands out in current peptide research.
Researchers do not usually frame the molecule as a general peptide in the same way as a broad catalog entry such as a signaling fragment or short experimental sequence. Instead, they discuss it as a highly specific receptor-active investigational compound with a defined mechanism being explored in structured studies.
Why the “triple agonist” label matters
A one-pathway design acts at one receptor family. A two-pathway design acts at two. Retatrutide is different because it combines three receptor signals in one molecule. That design has made it an important subject in obesity and liver-focused research, where investigators are looking at how combined receptor activity changes outcomes across multiple endpoints.
How retatrutide works
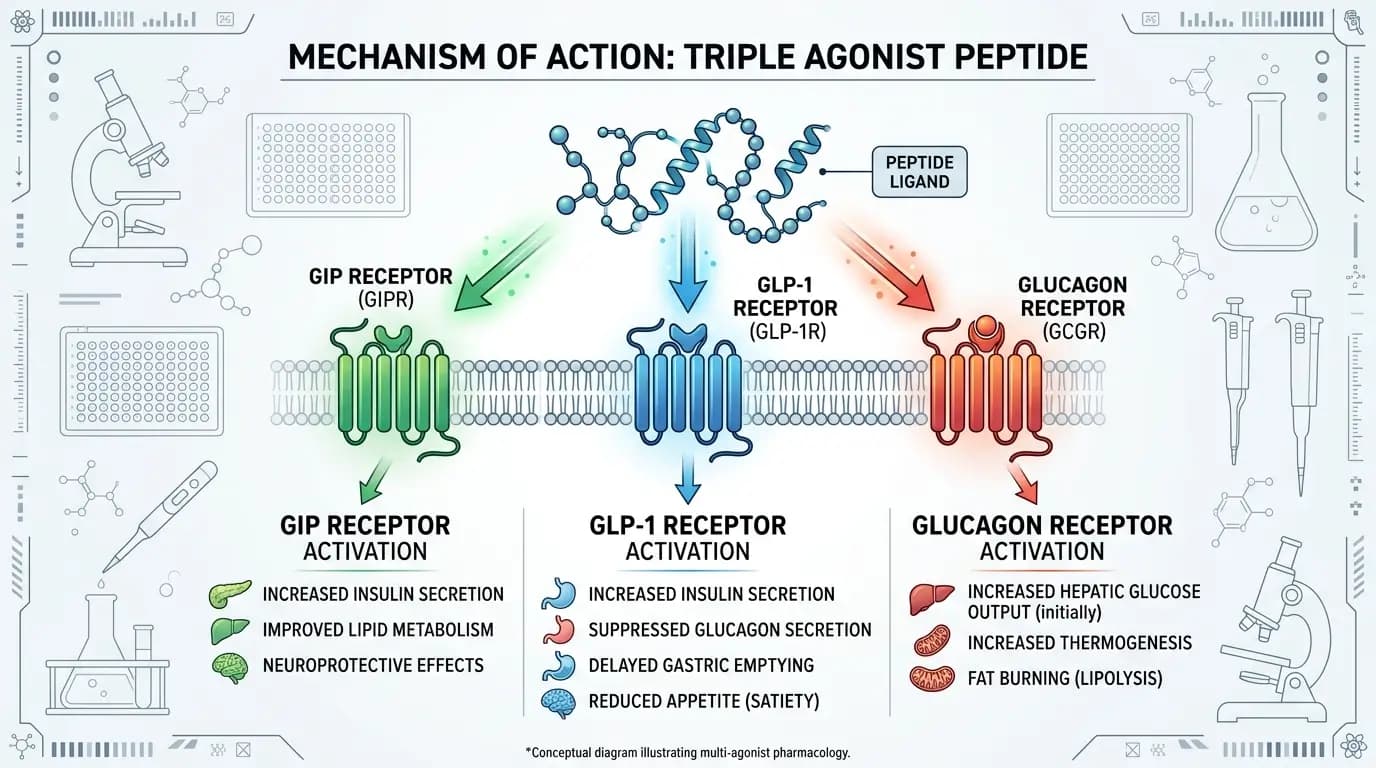
The three receptor pathways
The current research describes three receptor targets:
GIP receptor
GLP-1 receptor
Glucagon receptor
This matters because each pathway is tied to metabolic regulation, yet they are not identical in function. In broad terms, the triple-agonist concept aims to combine overlapping and complementary effects rather than rely on a single signaling route.
Why researchers find the design interesting
The logic behind this design is not just “more is better.” Instead, the goal is to study whether coordinated receptor activity produces a profile that differs from earlier single- and dual-pathway compounds. That is why papers and study programs keep emphasizing mechanism, comparative biology, and longer-duration follow-up.
A quick comparison table
Research feature | Single-pathway compounds | Dual-pathway compounds | Retatrutide |
|---|---|---|---|
Receptor targets | 1 | 2 | 3 |
Core signaling focus | Narrower | Broader | Broadest of the three |
Current research interest | Established | Strong | Very strong |
Status | Varies by compound | Varies by compound | Still investigational |
The table is a simplified way to understand why retatrutide receives so much attention. It is not a ranking of quality, and it does not prove superiority across compounds.
What published human research has shown so far
The Phase 2 trial
The most cited published human study is a 48-week randomized Phase 2 trial involving 338 adults. The primary endpoint was percentage change in body weight at 24 weeks. Secondary endpoints included the same measure at 48 weeks and threshold-based reductions of 5%, 10%, and 15% or more.
In that paper, least-squares mean percentage change in body weight at 24 weeks ranged from -7.2% in the 1 mg group to -17.5% in the 12 mg group, compared with -1.6% in the placebo group. At 48 weeks, the corresponding changes ranged from -8.7% to -24.2%, compared with -2.1% for placebo. The paper also reported that gastrointestinal events were the most common adverse events and that dose-dependent increases in heart rate peaked at 24 weeks and then declined.
What those numbers mean
These results explain why the molecule moved quickly into larger late-stage programs. Still, one published Phase 2 trial does not answer every question. It gives an important signal, yet it does not replace the need for larger programs, broader participant populations, and longer follow-up.
Liver-focused findings
A separate published substudy in Nature Medicine examined liver-fat endpoints in participants with metabolic dysfunction-associated steatotic liver disease and at least 10% liver fat. After 24 weeks, relative liver-fat reduction was significantly greater than placebo across all studied dose groups, with the largest published reductions in the 8 mg and 12 mg groups. That paper added another reason researchers keep following retatrutide beyond one headline endpoint alone.
What is happening now
Phase 3 development
The late-stage TRIUMPH program is designed to evaluate retatrutide across more than 5,800 participants in four Phase 3 studies. According to the published design paper, these trials examine obesity-related endpoints and also include linked work involving obstructive sleep apnea, knee osteoarthritis, and cardiovascular-risk populations.
ClinicalTrials.gov also lists active studies involving retatrutide in obesity and cardiovascular-risk settings, showing that the program is still moving through formal evaluation rather than sitting at a completed, approved stage.
Why that stage matters
Phase 3 work matters because it is meant to test whether earlier signals remain consistent at larger scale. It also helps clarify tolerability, durability, and performance across different participant groups. Until that body of evidence is complete and reviewed by regulators, any discussion should stay grounded in published data rather than internet hype.
Is retatrutide approved?
Retatrutide is still investigational. FDA warning and compounding documents from 2025 and 2026 state that products containing retatrutide sold online were unapproved and that retatrutide is not identified as an active ingredient in any FDA-approved drug. FDA also states that companies have falsely labeled products containing retatrutide as being for research purposes or not for human consumption while selling them directly to consumers.
That status point is essential. A compound can generate strong research interest and still remain outside approved commercial status. Those two facts are not contradictory. They simply describe different stages in the evidence and regulatory pathway.
Why online listings create confusion
Research labels can be misleading
One of the biggest problems around this topic is language. FDA has warned that some online sellers used research-style labels while also presenting products in ways that indicated intended human use. The agency says such products may be counterfeit, contain the wrong ingredients, contain harmful ingredients, or contain too little, too much, or no active ingredient at all.
For a reader trying to understand the subject, this means the phrase “research” on a webpage does not automatically mean the overall page is actually operating as a neutral research resource. Intent is inferred from the full page, not from one disclaimer alone. That is also why neutral scientific language matters.
What careful readers should check
When reading any page about retatrutide, it helps to check:
whether the page clearly says the compound is investigational
whether the claims match published papers
whether the page avoids consumer outcome promises
whether it separates study findings from sales language
whether it cites formal sources such as PubMed, Nature Medicine, NEJM, ClinicalTrials.gov, or FDA pages
Retatrutide compared with related compounds
Is it the same kind of molecule as dual agonists?
No. Mechanistically, retatrutide differs from dual agonists because it adds glucagon receptor activity to the GIP and GLP-1 receptor profile. That does not automatically prove it is “better” in every research context, but it does explain why papers regularly describe it as a next-generation multi-receptor design.
Why simple rankings can be misleading
Readers often want a quick winner between one compound and another. Real research is rarely that simple. Cross-trial comparisons can be misleading because study populations, durations, endpoints, and escalation methods differ. A clean answer usually requires a direct head-to-head study, and that is not the same as comparing headline numbers from different papers or websites. The safest reading is that retatrutide is mechanistically distinct and currently very important in ongoing research, while final comparative judgments still need fuller evidence.
Key limits in the evidence
Early strength does not end the story
The existing published data are meaningful, but there are still open questions around durability, longer follow-up, broader populations, and how outcomes vary across different subgroups. Even strong early numbers must be placed inside the context of program stage and study scope.
Public discussion often outruns the data
Some online pages move too quickly from “interesting results” to “finished conclusion.” That is a mistake. The proper scientific reading is narrower: the molecule has shown strong signals in published work, larger studies are underway, and regulatory review is not complete.
How to read retatrutide research well

Start with the study model
First, identify whether the source is a human study, animal work, a review article, a trial listing, or a commercial page. These are not interchangeable. A formal paper and a sales page do not carry the same evidentiary weight.
Look at duration and endpoints
A short-duration paper may reveal an early signal, but a longer paper can show whether the effect continues, slows, or changes over time. Endpoint choice also matters. One paper may focus on body weight, another on liver fat, and another on broader cardiometabolic measures.
Separate published findings from promotional language
The safest approach is to prefer peer-reviewed publications, PubMed records, ClinicalTrials.gov entries, and FDA pages. Those sources are not hype-free in every line, but they are far stronger than recycled blog content or social posts.
Conclusion
Retatrutide is an investigational triple-agonist peptide that has become a major subject in current metabolic research. Its defining feature is simultaneous activity at GIP, GLP-1, and glucagon receptors. Published Phase 2 work and liver-focused data explain the strong interest, while ongoing Phase 3 programs show the story is still unfolding. FDA materials also make clear that online sales language around this compound can be misleading and that unapproved products sold under research-style labels raise major concerns. The most accurate takeaway is simple: retatrutide is one of the most closely watched molecules in this area, but it remains a research-stage compound whose final place depends on the evidence still being generated.