For laboratory research use only. This article discusses peptides in a scientific and educational context.
Peptides are short chains of amino acids that appear throughout biology and laboratory research. In simple terms, when people ask what are peptides, they are asking about small molecular sequences built from amino-acid units linked by peptide bonds. This topic matters because peptides sit at the intersection of chemistry, biochemistry, and molecular research, and they help explain how larger biomolecules are assembled and studied.
When beginners search what are peptides, they often see peptides described as “small proteins.” That shortcut can be useful at a basic level, but it is incomplete. Peptides and proteins are related, yet they are not always described in exactly the same way, because size, structure, and folding all influence how researchers classify them.
What Are Peptides in Simple Terms

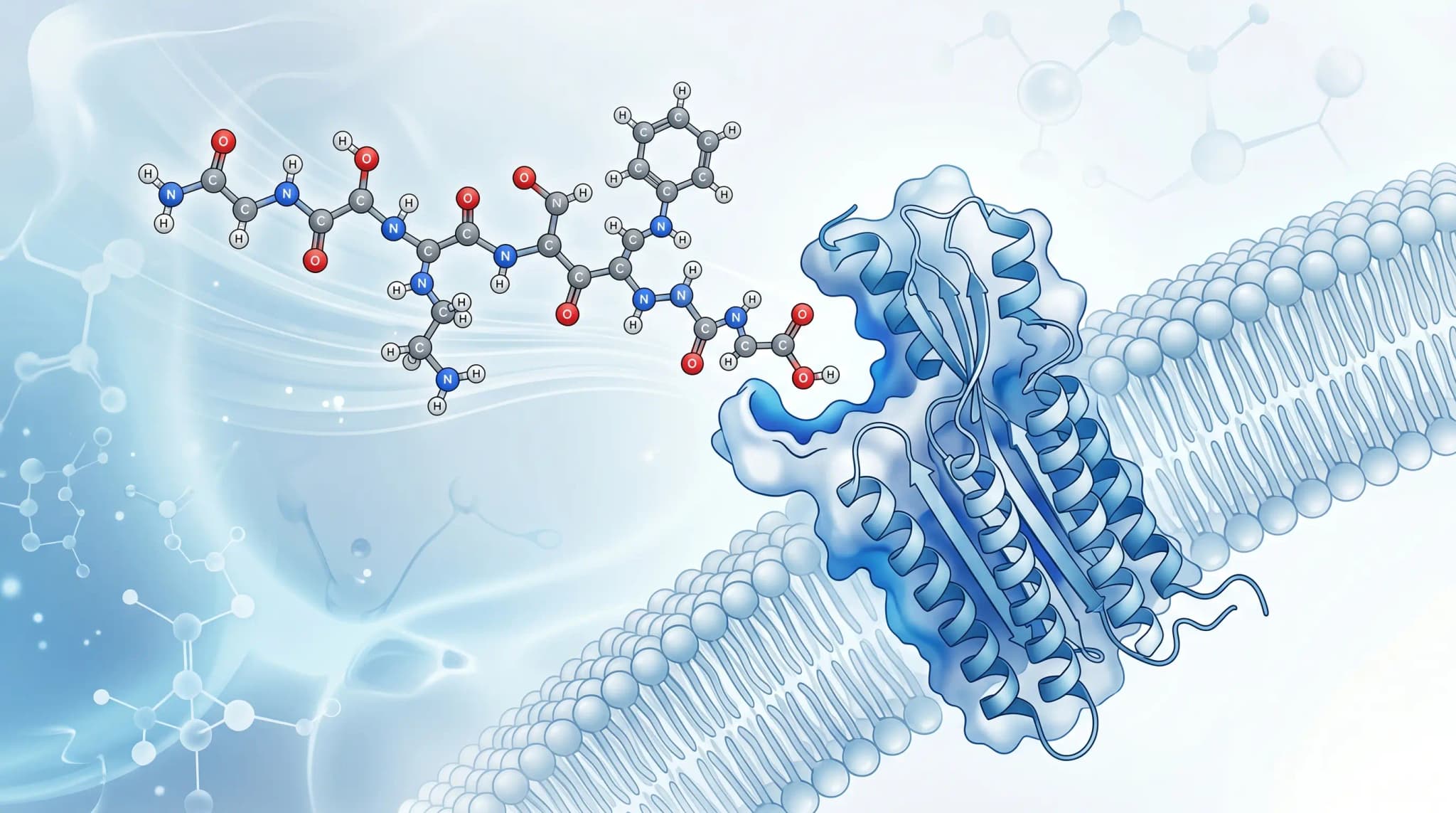
The clearest answer to what are peptides is this: peptides are short chains of amino acids joined by peptide bonds. The National Human Genome Research Institute describes a peptide as a short chain, typically 2 to 50 amino acids long. In that same framework, a longer chain is usually described as a polypeptide, and proteins are made from one or more polypeptides.
Amino acids are often called the building blocks of proteins. Each amino acid contains a common backbone plus a side chain, and those side chains influence how a peptide behaves in water, near membranes, or around other molecules. Because of that, even a short sequence can have very different chemical properties from another sequence of the same length.
Another useful way to answer what are peptides is to think in terms of sequence. A peptide is not just a random cluster of atoms. Instead, it is an ordered chain, which means the position of each amino-acid residue matters. Even a one-residue change can alter charge, shape, solubility, or binding behavior in a research setting.
What Is a Peptide Bond
A peptide bond is the chemical link that joins one amino acid to the next. Britannica explains that this bond forms between the carbon atom of one amide group and the nitrogen atom of the next, creating the backbone that runs through the chain. That backbone is central to how peptides are named, drawn, and analyzed.
The peptide bond is not a loose connection. NCBI notes that it has partial double-bond character, which makes it more rigid and planar than an ordinary single bond. As a result, the peptide backbone has structural constraints, and those constraints help determine how a peptide can twist, bend, or assemble.
Why Size Matters
When people ask what are peptides, they are often really asking where peptides end and proteins begin. There is no single universal cutoff used in every field. Still, many educational sources use a practical range in which peptides contain about 2 to 50 amino acids, oligopeptides are shorter subsets, and proteins are larger or more structurally complex chains.
That distinction is helpful, although it is not perfectly rigid. Some sources emphasize molecular size, whereas others emphasize how extensively a chain folds into stable three-dimensional forms. In research writing, both ideas appear, which is why the language around peptides, polypeptides, and proteins sometimes overlaps.
How Peptides Are Formed
To understand what are peptides, it helps to know how they form. NCBI describes peptide formation as a condensation reaction in which amino acids become linked through covalent bonding. During that process, the reaction removes water, and scientists call the remaining portion of each amino acid in the chain a residue.
In cells, peptide formation is tied to translation at the ribosome. As amino acids link one after another, a growing chain emerges with a specific sequence. Eventually, that chain may remain relatively short as a peptide, or it may extend into a longer polypeptide that later folds into a protein.
Researchers usually describe a peptide from the N-terminus to the C-terminus. In other words, the chain has a directional order, and that order matters when scientists compare sequences, discuss substitutions, or interpret analytical data. Therefore, peptide notation is not just a naming habit, it is part of how sequence identity is communicated.
Linear Peptides and Cyclic Peptides
Most beginners first encounter linear peptides, which have one end at the N-terminus and another at the C-terminus. These are the most straightforward to visualize, and they are common in introductory diagrams and sequence tables.
Cyclic peptides form a closed loop or contain ring-like constraints created through chemical linkage. Because ring formation changes flexibility and exposure of residues, the research literature often discusses cyclic peptides separately from linear ones. In practice, that difference can affect conformation, stability, and interaction patterns in experimental systems.
Peptides vs Proteins
A major reason people search what are peptides is to compare them with proteins. Structurally, both are made from amino-acid chains joined by peptide bonds. Britannica explains that the main difference is usually size and structural complexity, with peptides being smaller and often less defined in overall structure than proteins.
Proteins commonly adopt secondary, tertiary, and sometimes quaternary structure. Peptides can form local structure too, but researchers often discuss them as shorter segments, fragments, motifs, or signaling units rather than large, highly folded macromolecules. Consequently, the word “peptide” often signals a narrower scale of analysis.
A useful mini-table makes this easier to see:
Feature | Peptides | Proteins |
|---|---|---|
Typical length | Often about 2 to 50 amino acids | Commonly larger chains or assemblies |
Bond type | Peptide bonds | Peptide bonds |
Structural complexity | Often simpler or more limited | Often more extensive folding |
Common research focus | Sequence motifs, binding regions, synthesis, analytical profiling | Enzymes, structural assemblies, multi-domain functions |
The table is a guide, not a rulebook. Overall, the boundary between peptide and protein depends on context, and some sources use the terms with slight variation.
Main Types of Peptides

Another practical way to answer what are peptides is to classify them by length, origin, and biological role. This helps beginners move from definition to real-world scientific categories.
Types by Length
By length, the names are fairly intuitive:
Dipeptide: two amino acids
Tripeptide: three amino acids
Oligopeptide: a short chain, often described as only a few to around 20 amino acids
Polypeptide: a longer chain that can extend toward protein-scale structure
These labels are widely used in education and research, although the exact boundary between oligopeptide and polypeptide can vary by source.
Types by Origin
Peptides can arise in several ways:
Naturally occurring peptides, produced in cells and organisms
Protein-derived peptides, released when larger proteins are cut into smaller fragments
Synthetic peptides, assembled in the laboratory for research and analytical work
This classification matters because origin influences purity targets, modification patterns, and the type of data researchers expect to collect. For example, a synthetic sequence may be built to test a hypothesis about binding or folding, whereas a naturally occurring peptide may be isolated to study its native role.
Types by Biological Role
Peptides can participate in many biological processes. Britannica notes that the class includes molecules such as hormones and antibiotics, and educational sources also describe peptides as important signaling and regulatory components in cells. Researchers often group peptides by function, for example as signaling peptides, regulatory peptides, structural motifs, or peptides involved in interactions with microbes.
That does not mean every peptide belongs neatly to one box. On the contrary, researchers may study a single sequence for several reasons at once, depending on the model, the assay, and the question they are testing.
Where Peptides Are Found
When people ask what are peptides, they sometimes imagine a narrow laboratory category. In reality, peptides are found throughout living systems. They appear in cells, tissues, microorganisms, plants, and animals, because amino-acid chains are basic components of biological organization.
Cells synthesize some peptides directly as short active sequences. Others appear only after a larger precursor is processed into smaller parts. NCBI describes how some biologically active peptides arise from larger precursor forms that undergo further cleavage before reaching their final state.
Peptides can even be discussed in the context of food chemistry and protein breakdown. When researchers hydrolyze proteins, they release smaller fragments that they may then isolate, identify, or compare during laboratory analysis. Thus, peptides can be viewed both as native biomolecules and as fragments generated during controlled processing.
Why Peptides Matter in Biology and Research
The question what are peptides becomes more interesting once structure meets function. Peptides matter because short amino-acid sequences can participate in signaling, recognition, assembly, transport, and molecular interaction. Even very short chains can carry useful information about how residues contribute to binding or conformation.
Researchers often use peptides as simplified systems. For example, a short synthetic sequence can help isolate one binding region from a much larger protein, making an experiment easier to design and interpret. Similarly, researchers can use peptide fragments to map epitopes, model interfaces, or test how substitutions affect molecular behavior.
Peptides are important in early-stage and in vitro research because they allow tightly defined sequence design. However, findings from cell-based systems, biochemical assays, or animal models remain model-specific. Therefore, researchers should not describe a peptide that behaves one way in a controlled experiment as having the same behavior in every context.
How Peptides Are Studied in the Lab

A beginner asking what are peptides often needs a practical laboratory answer as well. In research settings, researchers commonly design, synthesize, purify, verify, store, and then test peptides in analytical or biochemical workflows.
Peptide Synthesis
NCBI and peptide-synthesis reviews describe solid-phase peptide synthesis, often shortened to SPPS, as a standard laboratory approach for assembling peptide chains step by step on a solid support. Researchers widely use this method because it allows precise sequence construction and makes it easier to introduce defined residues or modifications.
In a simple summary, researchers add protected amino acids in a controlled order until the target sequence is complete. Afterwards, researchers can cleave the peptide from the support and move it into purification and characterization steps. Finally, researchers check the analytical profile to confirm that the sample matches the intended sequence as closely as possible.
Purity and Analytical Verification
Purity matters in peptide research because impurities can complicate interpretation. Researchers commonly use reversed-phase HPLC for peptide isolation and purification and widely employ mass spectrometry to confirm molecular mass or track representative peptide signals in analytical workflows.
A typical research dossier may include sequence identity, molecular mass, chromatographic profile, and storage notes. In addition, researchers often review salt form, peptide content, and the presence of related fragments when comparing one batch with another.
Storage and Stability in Research Context
Peptides are not all equally stable. Sequence length, residue composition, terminal groups, oxidation sensitivity, moisture exposure, and repeated freeze-thaw events can all influence sample quality over time. Because of that, storage conditions are usually specified in technical documentation rather than guessed from sequence alone.
For beginners, the key idea is simple: the same word “peptide” covers many different molecules, and each one may require its own handling logic in a laboratory environment. In other words, there is no single storage rule that fits every sequence.
Common Peptide Terms Beginners Should Know
When learning what are peptides, a small glossary helps a lot.
Amino Acid
An amino acid is the molecular unit that makes up a peptide or protein. Standard amino acids share a core structure, but their side chains differ, and those differences influence charge, polarity, and reactivity.
Residue
Scientists often call an amino acid a residue once it becomes part of a peptide chain. NCBI explains this terminology in relation to the loss of water during condensation.
Sequence
Sequence refers to the order of residues in the chain. Because order determines chemical character and molecular recognition, sequence is one of the most important descriptors in peptide research.
Conformation
Conformation describes the three-dimensional arrangement adopted by a peptide. Some peptides remain flexible, whereas others prefer more ordered shapes under certain conditions.
Polypeptide
A polypeptide is a longer amino-acid chain. Depending on context, researchers may discuss it as a large peptide, a protein precursor, or part of a finished protein assembly.
Common Misunderstandings About Peptides
A common misunderstanding is that peptides and proteins are completely separate categories. In fact, they exist on a continuum of amino-acid chain length and structural complexity. Thus, the difference is real, but it is not a wall.
Another misunderstanding is that all peptides are naturally occurring. Many peptides studied in research are synthetic, precisely because laboratory synthesis allows control over sequence design, labeling, and modifications.
It is equally easy to assume that all peptides behave in the same way in solution. However, solubility, aggregation tendency, and stability depend on sequence and context. A hydrophobic peptide, for instance, may behave very differently from a charged peptide of similar length.
What Are Peptides in Research Practice

At this point, the answer to what are peptides One can summarize the findings from a laboratory perspective. Peptides occur naturally as short, sequence-defined amino-acid chains, or researchers assemble them synthetically for research. Researchers study them because they are chemically tractable, structurally informative, and useful for examining how sequence relates to molecular behavior.
For a beginner, that is the most practical takeaway. Peptides are small enough to study in a focused way, yet rich enough to reveal important ideas about chemistry, sequence, folding, and molecular interaction. As a result, they remain central tools in many areas of biochemical and analytical research.
Conclusion
Peptides are short chains of amino acids linked by peptide bonds, and that basic definition is the foundation for everything else in this topic. From there, the next important ideas are sequence order, chain direction, peptide bonds, size-based classification, and the relationship between peptides, polypeptides, and proteins.
For beginners, the best next step is to learn how amino-acid properties influence charge, polarity, and conformation. Then, move on to peptide synthesis, chromatographic analysis, and mass-based verification, because those are the tools that turn a definition into practical research understanding. Overall, once you understand what are peptides, many larger topics in molecular science become easier to follow.