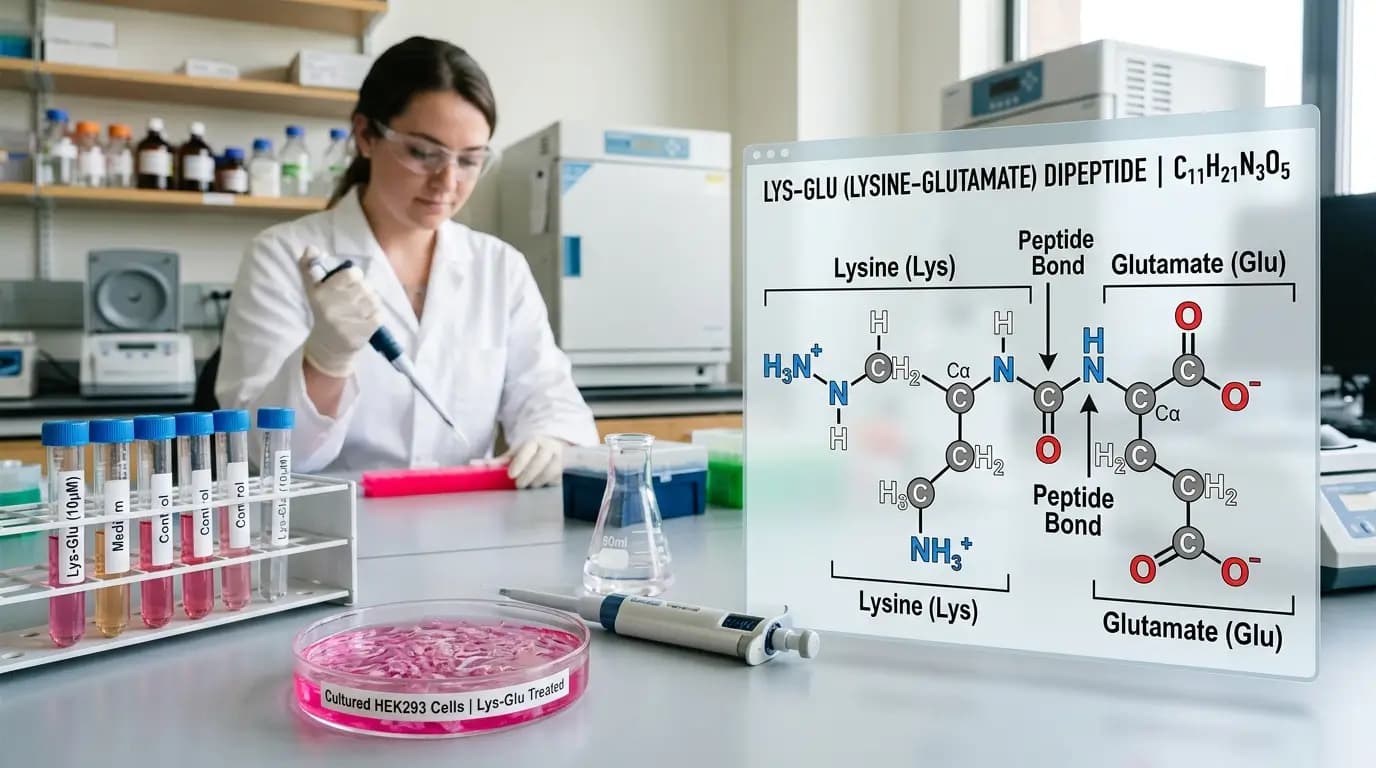
Vilon peptide appears in a compact but recurring set of molecular and cell-based papers. PubChem lists the compound as lysylglutamic acid, or Lys-Glu, with formula C11H21N3O5 and molecular weight 275.30 g/mol. In the literature, researchers usually place it within the short bioregulator peptide category and study it in defined culture models rather than in broad whole-system summaries.
For beginners, the topic can seem harder than it is. Many pages mix chemistry, cell biology, and oversized claims in the same space. This article takes a narrower route. It explains what vilon peptide is, which models appear most often, what mechanisms researchers discuss, and where the limits of the evidence start to matter. Overall, the strongest source trail centers on cultured lymphocytes, mouse thymocytes, pineal immune-cell culture, and THP-1 monocyte or macrophage systems.
What Is Vilon Peptide?

The simplest starting point is chemistry. Vilon peptide is the common name for lysylglutamic acid, a dipeptide built from lysine and glutamic acid residues. PubChem also lists the short form Lys-Glu, which helps when searching papers and product catalogs. Because the sequence is so short, researchers often discuss vilon peptide in the same family as other short peptide bioregulators.
Names matter here. Some pages use the common name, while others use the chemical name or the short sequence label. Therefore, a useful literature search usually includes all three. That small step makes it easier to connect older chromatin papers, thymocyte studies, and newer THP-1 work that might otherwise look unrelated at first glance for vilon peptide.
Core Molecular Profile
A basic profile helps readers place the molecule before moving into mechanism or model design.
Item | Detail |
|---|---|
Common name | Vilon peptide |
Alternate name | Lysylglutamic acid |
Short form | Lys-Glu |
Molecular formula | C11H21N3O5 |
Molecular weight | 275.30 g/mol |
Composition | Lysine + glutamic acid |
This profile looks simple, yet it matters. Researchers use these identifiers to confirm that different pages refer to the same compound in vilon peptide research. In addition, the very small size of the dipeptide partly explains why the literature often focuses on chromatin state, gene-access changes, and cell-culture behavior rather than on complex folding or large-structure analysis.
Why Researchers Study Vilon Peptide
The published work does not point to one single theme. Instead, the same few topics appear again and again across the source set. Those topics include chromatin organization, ribosomal gene activity, lymphocyte behavior, thymocyte proliferation, immune-cell differentiation, and inflammatory-pathway modulation in THP-1 models for vilon peptide.
A beginner can think of the field in three research lanes:
chromatin and gene-access papers
thymocyte and lymphocyte papers
monocyte or macrophage cell-line papers
This three-part view is useful because it keeps the discussion anchored to actual experiments. However, it also shows the narrowness of the evidence base. The literature is not large, and the same model families return frequently.
Chromatin-focused interest
One of the most cited vilon peptide papers examined cultured lymphocytes from older donors and described deheterochromatinization, reactivation of ribosomal genes, and release of genes repressed in facultative heterochromatin. Because of that paper, chromatin language now sits at the center of many summaries. Moreover, later pages often repeat those terms even when they do not explain the underlying model carefully.
Immune-cell interest
Another research lane links vilon peptide to thymocytes, lymphocytes, and related immune-cell systems. In mouse thymocytes, one paper reported a pronounced comitogenic effect on proliferation and changes associated with the sphingomyelin pathway. In organotypic pineal immune-cell culture, another paper described differentiation of precursor populations. Together, those studies explain why immune-cell language follows the compound across many current pages.
How the Literature Is Structured

The vilon peptide literature is small, but it still shows a recognizable pattern. Earlier papers focus on chromatin, lymphocytes, thymocytes, and nucleolar organizer regions. Newer work expands the discussion by using THP-1 cells, a human monocytic cell line that can differentiate into macrophage-like cells in vitro. Consequently, current summaries usually pull from two source buckets at once: older bioregulator papers and newer pathway-oriented cell-line studies.
Older papers
In the older vilon peptide literature, authors often compared several short peptides in the same experimental frame. They looked at chromatin state, blast transformation, differentiation, and related indicators of cell activity. For example, one paper reported that vilon peptide promoted thymocyte transformation into proliferating blast cells and changed argyrophilic proteins in nucleolar organizer regions. That kind of endpoint tells readers something important. Most early papers stay close to cell behavior and molecular markers, not broad system-wide conclusions.
Newer THP-1 work
A 2022 paper used THP-1 cells to examine several short peptides in a more modern in vitro framework. The authors reported modulation of proliferative patterns and reduced expression of TNF and IL-6 in differentiated, LPS-stimulated THP-1 cells. Because this design uses a familiar monocyte or macrophage model, it helps modern readers understand where vilon peptide fits in pathway-oriented research. Still, it remains a cell-line study, so the interpretation must stay inside that model.
Proposed Mechanisms
When papers discuss vilon peptide mechanism, the same terms return with notable consistency. These terms include heterochromatin, nucleolar organizer regions, ribosomal genes, sphingomyelinase activity, tyrosine phosphorylation, and cytokine expression. Although the exact language changes from paper to paper, the shared idea stays similar. Researchers investigate whether this short peptide can shift cell-state regulation in culture.
Chromatin and gene-access framing
The chromatin framing for vilon peptide comes mainly from the 2004 lymphocyte paper. In that study, authors described progressive activation of facultative heterochromatin with age-linked context and linked the shift to ribosomal gene reactivation. In other words, the paper did not present a simple one-step receptor story. Instead, it framed the molecule as part of a broader gene-access discussion inside cultured lymphocytes.
Signaling and inflammatory-pathway framing
The signaling framing comes from thymocyte and THP-1 studies. In mouse thymocytes, vilon peptide correlated with stronger sphingomyelinase activity and proliferative responses than the other peptides tested in that paper. In the THP-1 study, it appeared in a panel that modulated tyrosine phosphorylation and reduced expression of TNF and IL-6 after inflammatory stimulation. By comparison, this lane gives the molecule a more contemporary pathway-centered presentation.
Vilon Peptide in Immune-Cell Research

Immune-cell research is one of the clearest reasons vilon peptide continues to attract interest. However, the phrase can sound wider than the evidence base really is. The source set does not cover every immune-cell category evenly. Instead, the literature concentrates on thymocytes, cultured lymphocytes, pineal immune-cell preparations, and THP-1 systems.
Thymocyte models
In the thymocyte paper, vilon peptide showed the strongest comitogenic effect among the short peptides compared in that experiment. The authors also linked it to sphingomyelinase activity in thymocyte membranes. Because the model used mouse thymocytes, the paper works best as a mechanistic reference point. It does not, by itself, define the molecule across all other settings.
Pineal immune-cell differentiation
The pineal vilon peptide paper offers a different angle. In organotypic culture, the authors reported differentiation of precursor cells into T-helper, cytotoxic T lymphocyte, and B-cell populations. This matters because it explains why some summaries describe the molecule in differentiation language rather than only in proliferation language. Moreover, it shows how the same compound can appear in closely related but not identical immune-cell questions.
THP-1 monocyte and macrophage models
The THP-1 study matters for a practical reason. Many readers already know that model, so the paper gives the field a more familiar anchor. There, the peptide appeared in a multi-peptide panel that modulated proliferative markers and reduced TNF and IL-6 expression after LPS stimulation in differentiated cells. Accordingly, newer discussions often connect the compound with inflammatory-pathway work in macrophage-like settings.
Vilon Peptide and Age-Related Research
A large share of current page copy wraps vilon peptide in strong age-related language. The careful reader should separate that page copy from the actual papers. The core published basis is narrower. One major paper examined cultured lymphocytes from older donors and described chromatin activation, while a later paper grouped this compound with other short peptides in chromatin work involving older individuals. That source trail is why vilon peptide appears so often in cellular aging discussions.
At the same time, the vilon peptide literature does not justify turning the topic into a catch-all answer for every age-related question. The evidence remains model-specific, paper-specific, and limited in scale. Therefore, the safest reading is simple: the compound appears in age-related molecular research, especially where authors study chromatin state and immune-cell behavior in culture.
Comparing This Dipeptide With Other Short Peptides
Vilon peptide rarely appears alone. Researchers often compare it with Epitalon, Thymogen, Cortagen, Thymalin, Chonluten, or related short peptides. This matters because comparative papers help readers see how the field organizes compounds by sequence length, model choice, and pathway behavior instead of by one universal label.
One useful vilon peptide example comes from the 2006 chromatin paper. There, authors reported that Epitalon and Livagen affected pericentromeric structural heterochromatin, while this dipeptide did not show that exact effect. In the THP-1 paper, the molecule appears next to Thymogen and Chonluten under shared assay conditions, which makes cross-comparison easier. Together, those papers show why beginners should not treat all short peptides as interchangeable.
Technical Questions Researchers Commonly Check

Before moving into claims, vilon peptide researchers usually ask a smaller set of technical questions. Those questions often reveal more than page-level copy does.
Common checkpoints
confirm that the common name, chemical name, and Lys-Glu label all refer to the same compound
note the formula and molecular weight
identify whether the paper uses lymphocytes, thymocytes, pineal culture, or THP-1 cells
separate cell-culture findings from broader interpretation
check whether the paper studies chromatin, proliferation, differentiation, or cytokine expression
read comparison studies closely when multiple peptides appear in one assay
These checkpoints are valuable because the vilon peptide literature is compact. A small wording shift can change the meaning of a summary very quickly. For example, a statement from cultured lymphocytes does not automatically describe THP-1 behavior, and a finding in mouse thymocytes does not automatically define the compound in every later paper.
Solubility and sample preparation
Technical handling also needs careful wording. Sigma notes that no universal solvent works for all lyophilized peptides, while Thermo Fisher recommends testing solubility carefully and selecting water, buffer, or other solvents according to peptide properties and assay needs. Therefore, any preparation note should stay tied to the product sheet, the assay, and the composition of the sample rather than rely on a universal rule.
Limits of Current Vilon Peptide Research
The biggest vilon peptide limit is scale. The source base is not wide, and the same experimental families recur often. Most of the visible literature clusters around cultured lymphocytes, mouse thymocytes, pineal immune-cell preparations, and THP-1 cells. Because of that, readers should view the topic as interesting but narrow rather than broad and settled.
A second vilon peptide limit is interpretation drift. Some commercial pages stretch small-model findings into much larger narratives. Yet the underlying papers mostly measure chromatin state, cell differentiation, proliferative markers, or cytokine expression in very specific systems. In contrast, broad page-level claims often move past the source material. Accordingly, a strong reading always identifies the model, the endpoint, and the boundary of the finding.
Conclusion
Vilon peptide is best understood as lysylglutamic acid, a short dipeptide that appears in a focused set of molecular and cell-based studies. The clearest recurring themes involve chromatin organization, ribosomal gene activity, thymocyte behavior, immune-cell differentiation, and inflammatory-pathway work in THP-1 models. Moreover, the safest summary is not that the topic answers every broad question, but that it remains a compact research subject with a few recurring mechanistic lanes.
For beginners, the next step is practical. Check the alternate names, identify the exact model, and ask whether the claim stays inside the measured endpoint. With that approach, vilon peptide literature becomes easier to read, compare, and evaluate without drifting away from the published source set.