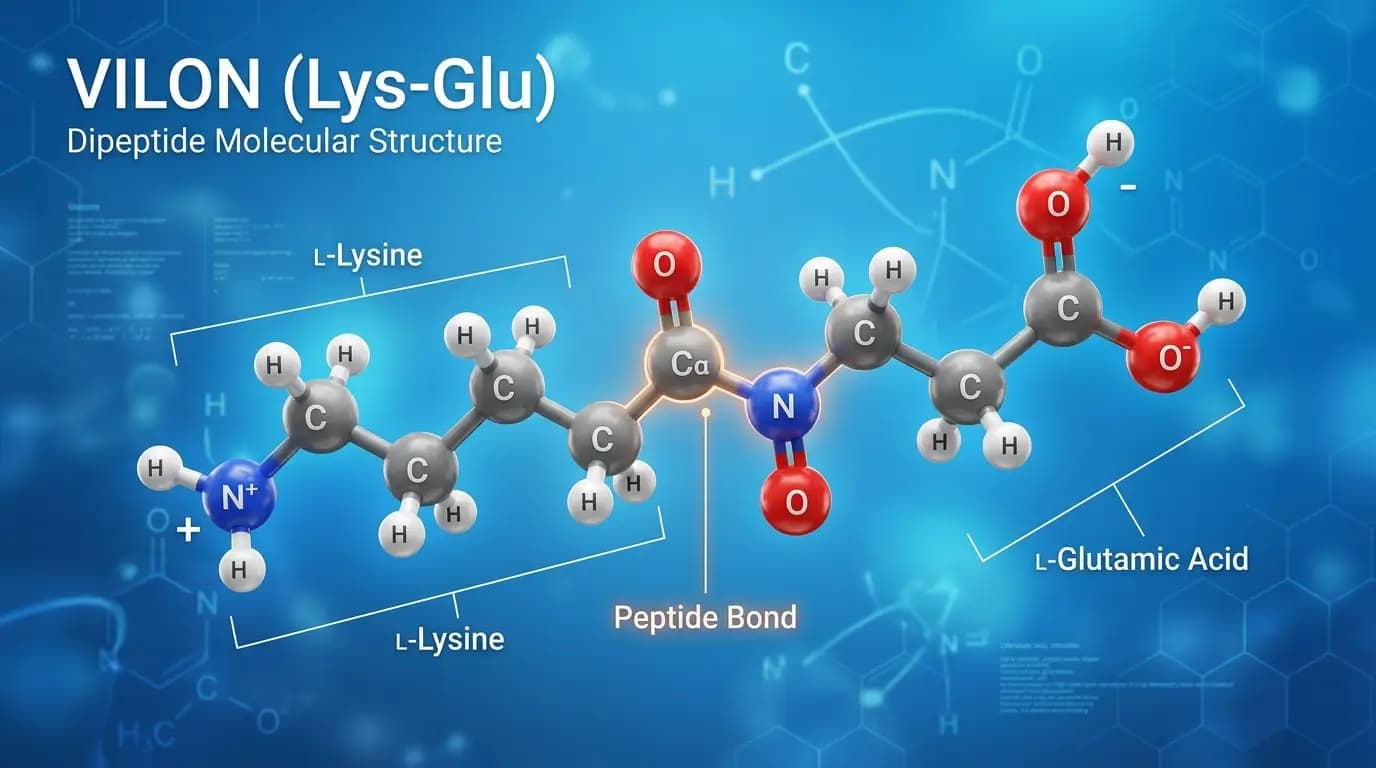
Vilon peptide represents a synthetic dipeptide consisting of the amino acids L-lysine and L-glutamic acid. Furthermore, molecular biology recognizes this molecule as one of the shortest signaling molecules capable of influencing cellular activity. Because it possesses a very low molecular weight, researchers often categorize it as a bioregulator. This specific dipeptide belongs to a class of substances originally developed to investigate how small molecules interact with the genetic apparatus of a cell. Consequently, vilon peptide has become a primary subject in studies focusing on chromatin structure and the regulation of gene expression. Scientists utilize this molecule in various laboratory models to understand the fundamental mechanisms of cellular resilience and signaling.
Molecular Identity and Chemical Specifications of Vilon Peptide

Its simple yet potent chemical structure defines the molecular identity of vilon peptide. Specifically, the molecule is a dipeptide, which means it contains only two amino acids linked by a single peptide bond. This structure allows the molecule to maintain high stability under specific laboratory conditions. Because the molecular weight is approximately $257.3$ g/mol, the molecule can easily penetrate cellular membranes in vitro. Therefore, the vilon peptide serves as an efficient tool for exploring intracellular signaling pathways.
Chemical Formula and Primary Structure
The chemical formula for vilon peptide is $C_{11}H_{21}N_{3}O_{5}$In a laboratory setting, researchers identify the primary structure as L-lysyl-L-glutamic acid, commonly abbreviated as Lys-Glu. This combination of a basic amino acid (lysine) and an acidic amino acid (glutamic acid) creates a unique electrostatic profile. Similarly, researchers believe this profile facilitates interactions between the peptide and negatively charged DNA molecules. Because of this specific arrangement, researchers often investigate the vilon peptide for its ability to bind to various regions of the genome.
Analytical Testing: HPLC and Mass Spectrometry Verification
To ensure the integrity of laboratory experiments, researchers must verify the purity of the vilon peptide. Specifically, High-Performance Liquid Chromatography (HPLC) is the standard method used to determine the chemical purity of the sample. In addition, Mass Spectrometry confirms the exact molecular mass and sequence of the dipeptide. Most research-grade batches of vilon peptide maintain a purity level exceeding 98%. Consequently, these rigorous testing methods prevent the interference of contaminants in cellular research models.
Solubility and Handling in Laboratory Settings
Vilon peptide is highly soluble in aqueous solutions, such as sterile water or phosphate-buffered saline. Because of its stability, researchers can store the lyophilized powder at -20°C for extended periods without significant degradation. However, researchers must handle the reconstituted solution with care to avoid bacterial contamination. Specifically, any work involving the vilon peptide should occur in a sterile environment, such as a laminar flow hood. In contrast to larger proteins, this dipeptide is less sensitive to mechanical agitation but remains susceptible to high temperatures.
Property | Specification |
Sequence | Lys-Glu |
Molecular Weight | $257.3$ g/mol |
Purity (HPLC) | >98% |
Physical Form | Lyophilized Powder |
Solubility | Water-Soluble |
Epigenetic Mechanisms: Chromatin Modulation and Gene Expression
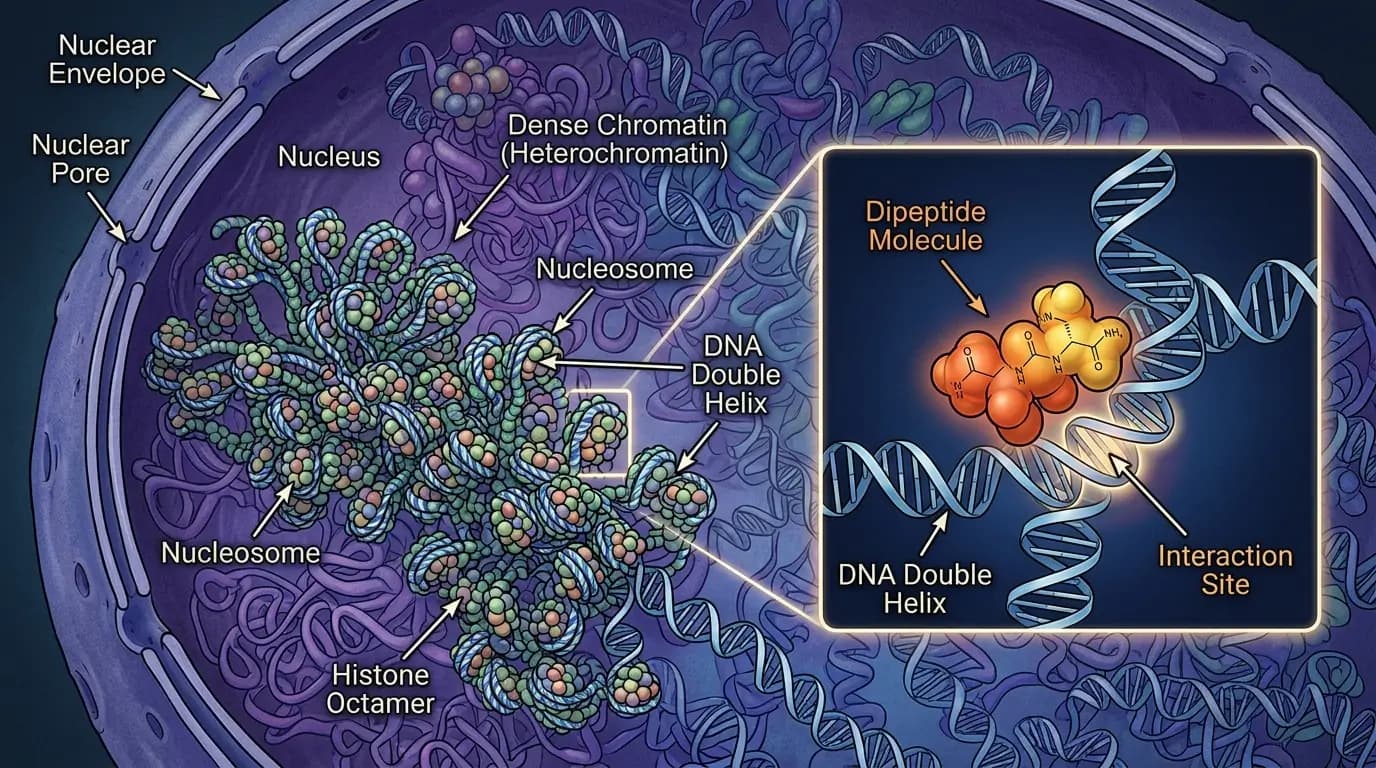
The interaction between vilon peptide and the cell nucleus is a cornerstone of modern peptide research. Specifically, studies suggest that this dipeptide can alter the physical state of chromatin. Therefore, researchers investigate how the molecule might "unlock" certain regions of the genome that are typically inaccessible. Because epigenetic regulation governs how cells respond to stress, understanding these mechanisms is crucial for molecular biology. Furthermore, researchers study vilon peptide for its potential to modulate the transcription of genes involved in cellular maintenance.
Interaction with Facultative Heterochromatin and DNA Binding
Research indicates that vilon peptide may promote the decondensation of facultative heterochromatin. This process, often referred to as chromatin unwrapping, allows the transcriptional machinery of the cell to access previously silenced genes. Specifically, researchers believe the dipeptide binds to the major groove of the DNA double helix. Similarly, this binding may displace certain histones or other repressive proteins. Because this interaction is highly specific, vilon peptide serves as a valuable molecular probe for studying the dynamics of the nuclear envelope and DNA architecture.
Activation of Ribosomal Genes and Nucleolus Organizer Regions (NORs)
In various cellular models, vilon peptide has demonstrated an ability to activate ribosomal genes. Specifically, researchers observe increased activity in the nucleolus organizer regions (NORs) of the cell. Because ribosomes are responsible for protein synthesis, this activation suggests a boost in the cellular capacity for maintenance. Furthermore, data from senescent cell cultures indicate that vilon peptide may help restore the activity of these regions to levels seen in younger cells. Consequently, this dipeptide is a significant focus in studies regarding cellular vitality at the genomic level.
Impact on Epigenetic Markers and Methylation Patterns
The vilon peptide is also investigated for its impact on DNA methylation patterns. Specifically, researchers examine whether the molecule can influence the activity of DNA methyltransferases. Because methylation is a primary mechanism for gene silencing, altering these patterns can have profound effects on cellular behavior. In addition, studies explore how the vilon peptide interacts with acetylated histones. Therefore, the dipeptide is considered a potential modulator of the "histone code," which dictates the accessibility of genetic information within the cell.
Preclinical Observations in Immunological Research
The immunological properties of vilon peptide are a frequent subject of preclinical investigation. Specifically, research models suggest that this dipeptide can influence the signaling pathways of the adaptive immune system. Because the thymus is the primary site for T-cell maturation, many studies focus on how vilon peptide interacts with thymic tissue. Similarly, researchers observe changes in the expression of various surface markers on immune cells. Consequently, researchers use the vilon peptide to explore the fundamental principles of immunomodulation in a laboratory context.
T-Lymphocyte Differentiation and Thymic Maturation Signaling
Studies involving thymic cell cultures suggest that vilon peptide may support the differentiation of T-lymphocytes. Specifically, the peptide influences the ratio of different T-cell subsets, such as helper and suppressor cells. Because these cells are critical for a coordinated immune response, understanding their maturation process is essential. Furthermore, researchers investigate how vilon peptide might signal to thymic epithelial cells to produce various cytokines. In addition, these studies often use flow cytometry to track the changes in cell populations over time.
Cytokine Expression and Inflammatory Pathway Investigations
Researchers frequently use Vilon peptide to study the expression of pro-inflammatory and anti-inflammatory cytokines. Specifically, in vitro models of inflammation show that the peptide may influence the production of Interleukin-6 (IL-6) and Tumor Necrosis Factor (TNF). Because these molecules are central to the inflammatory response, the vilon peptide provides a way to examine cellular signaling during stress. Furthermore, researchers investigate how the dipeptide might inhibit the activation of the NF-kappaB pathway. Therefore, the molecule is a valuable tool for understanding the molecular triggers of inflammation.
Impact on Lymphocyte and Granulocyte Counts in Stress Models
Research using murine models has examined how vilon peptide affects the concentration of white blood cells under various experimental conditions. Specifically, observations suggest that the peptide may help maintain lymphocyte and granulocyte counts during periods of physiological stress. Because these cells are the first line of defense, their stability is a key indicator of cellular resilience. Similarly, researchers analyze the bone marrow of these models to see if the vilon peptide influences the production of hematopoietic stem cells. Consequently, these findings contribute to a broader understanding of how small peptides support biological stability.
Investigations in Tissue Regeneration and Cellular Proliferation
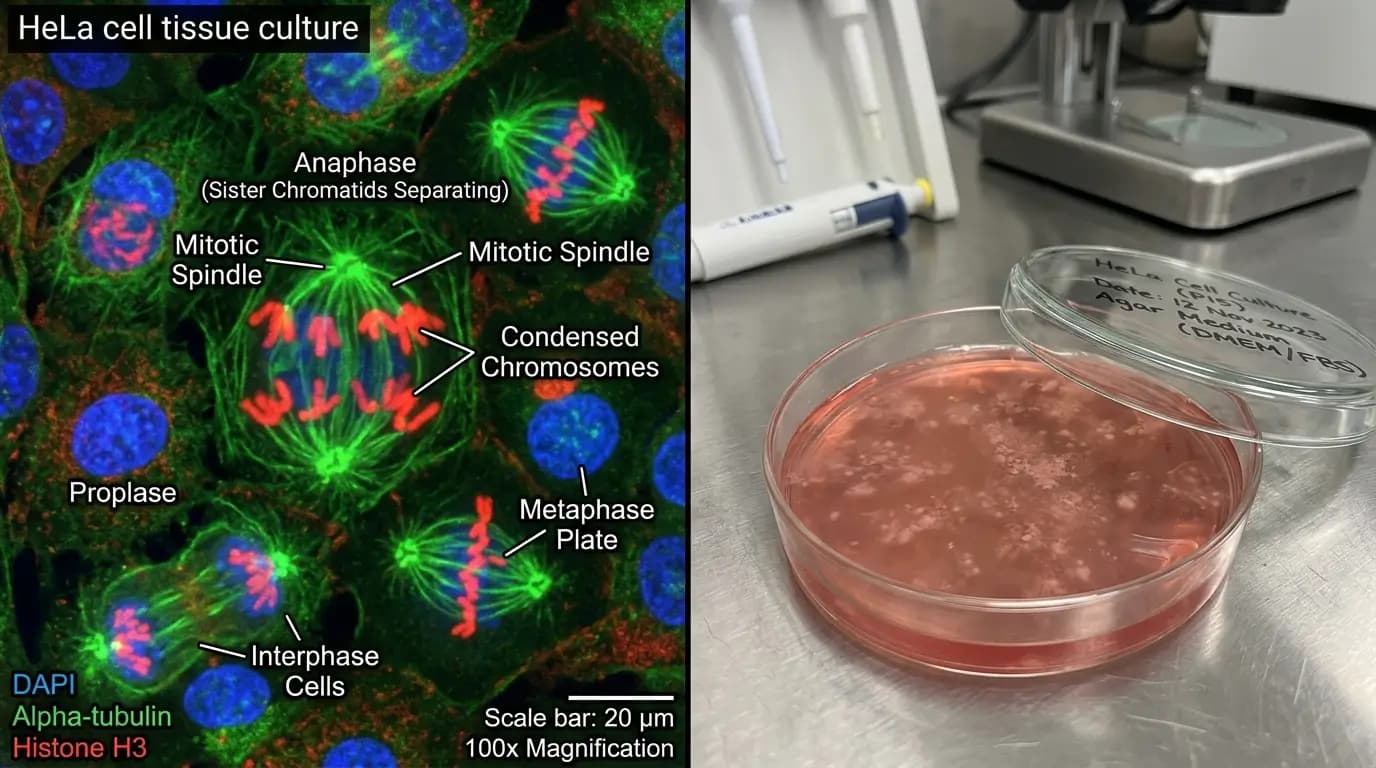
The role of vilon peptide in tissue regeneration is a growing area of scientific interest. Specifically, researchers use organotypic tissue cultures to observe how the dipeptide influences the rate of cell division. Because tissue repair requires a precise balance of proliferation and differentiation, these studies are highly complex. Furthermore, researchers investigate vilon peptide for its potential to stimulate the activity of various growth factors. Therefore, researchers frequently include the dipeptide in research protocols involving the study of wound healing mechanisms in vitro.
Mitotic Activity in Splenocytes and Thymus Explant Studies
In experiments involving spleen and thymus explants, vilon peptide increased the mitotic index. Specifically, this means that the peptide may promote a higher frequency of cell division in these tissues. Because the spleen and thymus are vital for immune function, these observations are particularly significant. Furthermore, researchers investigate whether this increased activity leads to a higher density of functional cells. Similarly, these studies often compare the effects of vilon peptide to other known bioregulators to establish its relative potency.
Intestinal Mucosal Models and Crypt Stem-Cell Activation
Vilon peptide is also studied for its impact on the intestinal mucosa. Specifically, research in murine models explores how the dipeptide might influence the proliferation of cells within the intestinal crypts. Because the gut lining constantly replaces itself, it serves as an excellent model for studying stem cell activity. Furthermore, researchers examine whether the vilon peptide supports the integrity of the intestinal barrier. Consequently, these investigations provide insights into the molecular pathways that govern the health of epithelial tissues.
Fibroblast Proliferation and Angiogenesis Research in Vitro
The study of vilon peptide extends to its interaction with fibroblasts and endothelial cells. Specifically, in vitro research examines whether the dipeptide can stimulate the proliferation of fibroblasts, which are responsible for producing collagen. Because collagen is essential for tissue structure, this research is relevant to the study of skin and connective tissue. In addition, researchers investigate the potential for vilon peptide to promote angiogenesis, or the formation of new blood vessels. Therefore, the dipeptide is a central component of studies focusing on the molecular basis of tissue repair and regeneration.
Research identifies vilon peptide as a stimulator of fibroblast activity in cell culture.
Studies investigate the impact of the dipeptide on the expression of Vascular Endothelial Growth Factor (VEGF).
Observations suggest that vilon peptide may influence the migration of endothelial cells during vessel formation.
Researchers use these models to understand how dipeptides might facilitate the structural recovery of damaged tissues.
Molecular Research on Vascular and Metabolic Homeostasis
Researchers investigate Vilon peptide for its role in maintaining vascular and metabolic balance within a laboratory setting. Specifically, researchers examine how the dipeptide influences the production of molecules that regulate blood flow and coagulation. Because metabolic efficiency links to vascular health, these studies often overlap. Furthermore, researchers study vilon peptide for its impact on cellular metabolism, particularly in relation to energy production. Therefore, the dipeptide serves as a multifaceted tool for exploring the complex interactions between different biological systems.
Coagulation Pathways and Endogenous Anticoagulant Expression
Research in murine models has explored how vilon peptide influences the coagulation system. Specifically, the dipeptide is associated with an increase in the expression of endogenous anticoagulants, such as antithrombin III and protein C. Because these molecules prevent the formation of inappropriate blood clots, their regulation is a critical area of study. Furthermore, researchers investigate how the vilon peptide might modulate the activity of the fibrinolytic system. Consequently, these findings contribute to the understanding of how small peptides might support vascular homeostasis under experimental conditions.
Glucose and Lipid Metabolism in Cellular Models
The impact of vilon peptide on metabolic pathways is another active area of investigation. Specifically, researchers examine how the dipeptide influences the expression of genes involved in glucose transport and lipid oxidation. Because metabolic dysfunction is a hallmark of many cellular stressors, the vilon peptide is used to study how cells maintain energy balance. Furthermore, data suggest that the dipeptide may influence the sensitivity of cells to various metabolic signals. Similarly, these studies often use adipocyte and hepatocyte cultures to observe the specific effects of the peptide on fat and sugar processing.
Impact on Microvessel Permeability and Endothelial Stability
Vilon peptide is also studied for its influence on the structural integrity of the microvasculature. Specifically, research focuses on how the dipeptide might affect the permeability of endothelial cells that line the blood vessels. Because excessive permeability can lead to inflammation and tissue damage, maintaining endothelial stability is vital. Furthermore, researchers investigate whether vilon peptide can influence the expression of junctional proteins that hold endothelial cells together. Therefore, the dipeptide is an important subject in research regarding the molecular maintenance of the vascular barrier.
Exploring Cellular Senescence and Longevity Research
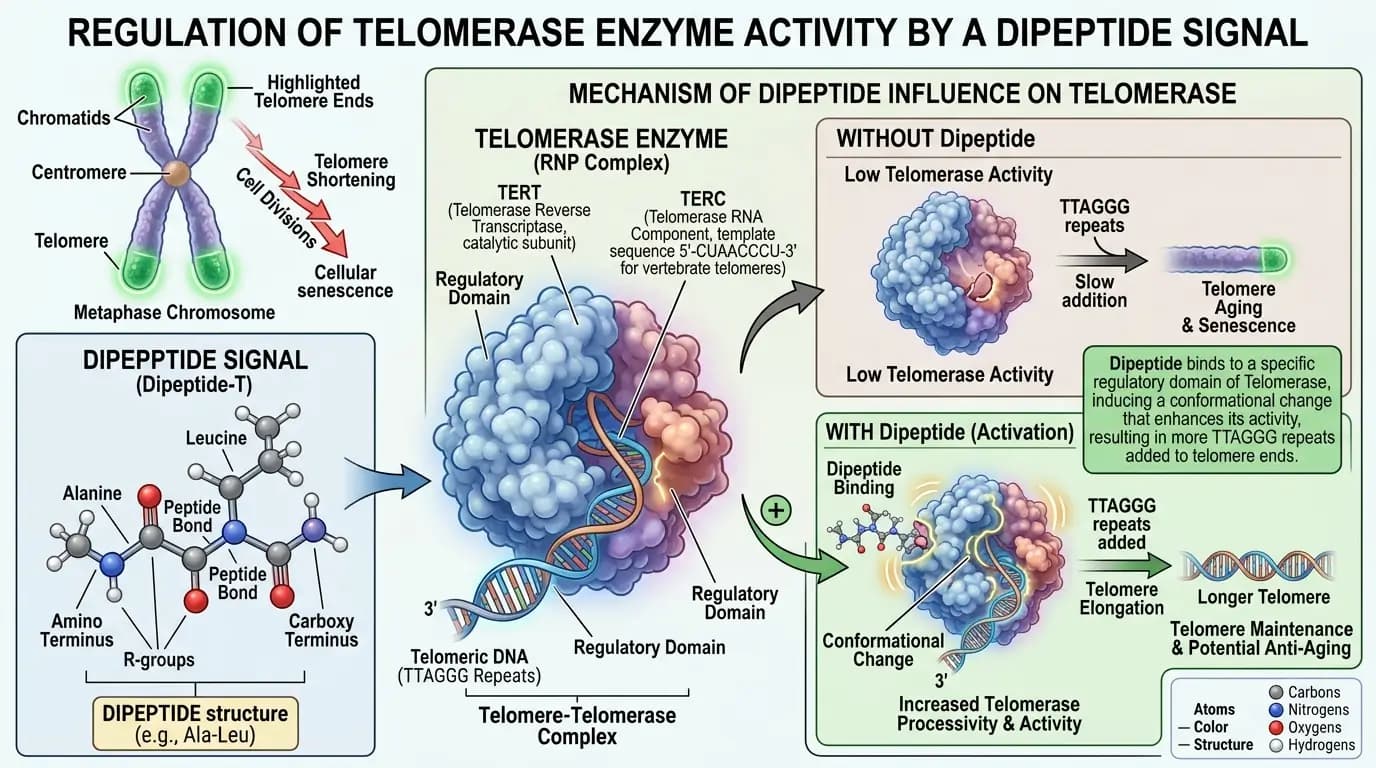
The vilon peptide is a prominent subject in the field of senescence research, where scientists study the processes of cellular aging. Specifically, researchers investigate how this dipeptide might influence the markers of biological age in various cell lines. Because senescence involves the gradual loss of cellular function, finding ways to observe and modulate this process is a major goal. Furthermore, vilon peptide is studied for its potential to extend the replicative lifespan of cells in vitro. Therefore, the dipeptide is central to the investigation of longevity at the molecular level.
Modulation of Telomerase Activity and Replicative Span
One of the most significant areas of vilon peptide research involves telomerase, the enzyme responsible for maintaining the ends of chromosomes. Specifically, studies examine whether the dipeptide can influence the expression of the TERT gene, which encodes the catalytic subunit of telomerase. Because telomere shortening is a primary cause of cellular senescence, modulating telomerase activity is a key focus. Furthermore, researchers have observed that cells treated with vilon peptide may undergo more divisions before reaching the Hayflick limit. Consequently, this dipeptide is used to explore the genomic factors that determine cellular longevity.
Observations in Murine Models Regarding Physiological Longevity
In long-term studies involving murine models, researchers have observed the impact of vilon peptide on overall physiological stability. Specifically, these experiments look at how the dipeptide affects the survival rates of the subjects over several months or years. Because these studies are conducted in a controlled environment, they allow researchers to isolate the effects of the peptide from external variables. Furthermore, the data often show changes in the expression of longevity-associated genes in these models. Similarly, these findings help scientists understand the broad impact of dipeptide signaling on the biological lifespan.
Interaction with Aging-Related Genes: FOXO1, TERT, and IGF1
Vilon peptide is investigated for its ability to interact with a specific set of genes associated with the aging process. Specifically, researchers look at the expression of FOXO1, which is involved in stress resistance and metabolism. Because these genes act as master regulators of cellular health, their modulation can have wide-ranging effects. In addition, studies examine the relationship between vilon peptide and the Insulin-like Growth Factor 1 (IGF1) signaling pathway. Therefore, the dipeptide is a valuable tool for mapping the complex genetic networks that govern how organisms age.
Comparative Analysis: Vilon vs. Other Bioregulatory Dipeptides
The scientific community often compares vilon peptide to other short-chain bioregulators, such as epitalon or thymogen. Specifically, while many of these peptides share a similar structural simplicity, their functional profiles can vary. Because each dipeptide has a unique amino acid sequence, it interacts with different molecular targets. Furthermore, researchers use these comparisons to determine the specific niche of vilon peptide within the landscape of peptide research. Consequently, this comparative approach helps refine the understanding of how different signaling molecules operate at the cellular level.
Vilon (Lys-Glu) is often contrasted with Epitalon (Ala-Glu-Asp-Gly) regarding their specific impact on telomerase activity.
Researchers compare the immunomodulatory effects of Vilon with those of Thymogen (Glu-Trp).
Studies investigate whether combining different peptides leads to synergistic effects in cellular models.
Comparative research helps identify the unique electrostatic interactions of the vilon peptide's Lys-Glu sequence.
Laboratory Compliance and Safety Protocols
Researchers working with vilon peptide must maintain strict adherence to safety and compliance protocols. Specifically, the substance is designated for laboratory research and development use only. Because the peptide is not intended for human consumption, all experiments must be restricted to in vitro and preclinical animal models. Furthermore, laboratories must ensure that all staff are trained in the proper handling of research peptides. Therefore, compliance with these rules is essential for maintaining the scientific validity and legal standing of the research.
Storage Stability and Degradation Prevention
To prevent the degradation of vilon peptide, it must be stored under optimal conditions. Specifically, the lyophilized powder should be kept in a moisture-free environment at temperatures between -20°C and -80°C. Because exposure to room temperature can lead to the breaking of peptide bonds, cold chain management is vital. Furthermore, once the peptide is dissolved, researchers should use it promptly or store it at 4°C for no more than 24 to 48 hours. Consequently, these storage protocols ensure that the experimental results are consistent and reproducible.
Ethical Research Practices and Non-Human Use Compliance
All research involving vilon peptide must follow established ethical guidelines for scientific investigation. Specifically, experiments involving animal models require approval from an Institutional Animal Care and Use Committee (IACUC). Because the vilon peptide is a research tool, it should never be represented as a medical product. Furthermore, researchers must accurately report all findings, including those that do not support their initial hypotheses. Therefore, maintaining high ethical standards is a fundamental requirement for anyone participating in peptide research.
Competitive sport organizations may restrict certain substances. Users are responsible for checking applicable rules.
Conclusion: The Future of Dipeptide Signaling Research
The vilon peptide remains a focal point of investigation due to its unique role as a short-chain signaling molecule. Because it can influence chromatin structure and gene expression, it provides a window into the most fundamental processes of cellular life. Specifically, the research conducted so far has highlighted its impact on immune signaling, tissue repair, and the markers of cellular senescence. Furthermore, as laboratory techniques continue to advance, scientists will be able to map the molecular interactions of the vilon peptide with even greater precision. Consequently, this dipeptide will likely continue to be an essential tool for researchers seeking to understand the complex pathways that govern biological stability and longevity.
References
Khavinson, V. Kh., et al. (2001). Tissue-Specific Effects of the Vilon Peptide (Lys-Glu) in Organotypic Cultures. Bulletin of Experimental Biology and Medicine.
https://pubmed.ncbi.nlm.nih.gov/11561563/Khavinson, V. K., & Malinin, V. V. (2005). Gerontological Aspects of Genome Peptide Regulation. Karger Publishers. DOI: 10.1159/isbn.978-3-318-01193-7
doi.org/10.1159/000084138Lezhava, T., et al. (2021). Epigenetic Modification Under the Influence of Peptide Bioregulators on Senescent Heterochromatin. PubMed Central.
https://pubmed.ncbi.nlm.nih.gov/33526740/Khavinson, V., et al. (2022). Peptides Regulating Proliferative Activity and Inflammatory Pathways in the Monocyte/Macrophage THP-1 Cell Line. MDPI International Journal of Molecular Sciences.
https://www.mdpi.com/1422-0067/23/7/3607Anisimov, V. N., et al. (2001). A Synthetic Dipeptide Vilon (L-Lys-L-Glu) and Observations of Biological Longevity in Murine Models. ResearchGate.
https://www.researchgate.net/publication/12375506_A_synthetic_dipeptide_vilon_L-Lys-L-Glu_inhibits_growth_of_spontaneous_tumors_and_increases_life_span_of_miceNational Center for Biotechnology Information. (2024). Compound Summary for Lys-Glu (Vilon Peptide). PubChem Database.
https://pubchem.ncbi.nlm.nih.gov/compound/Lys-Glu