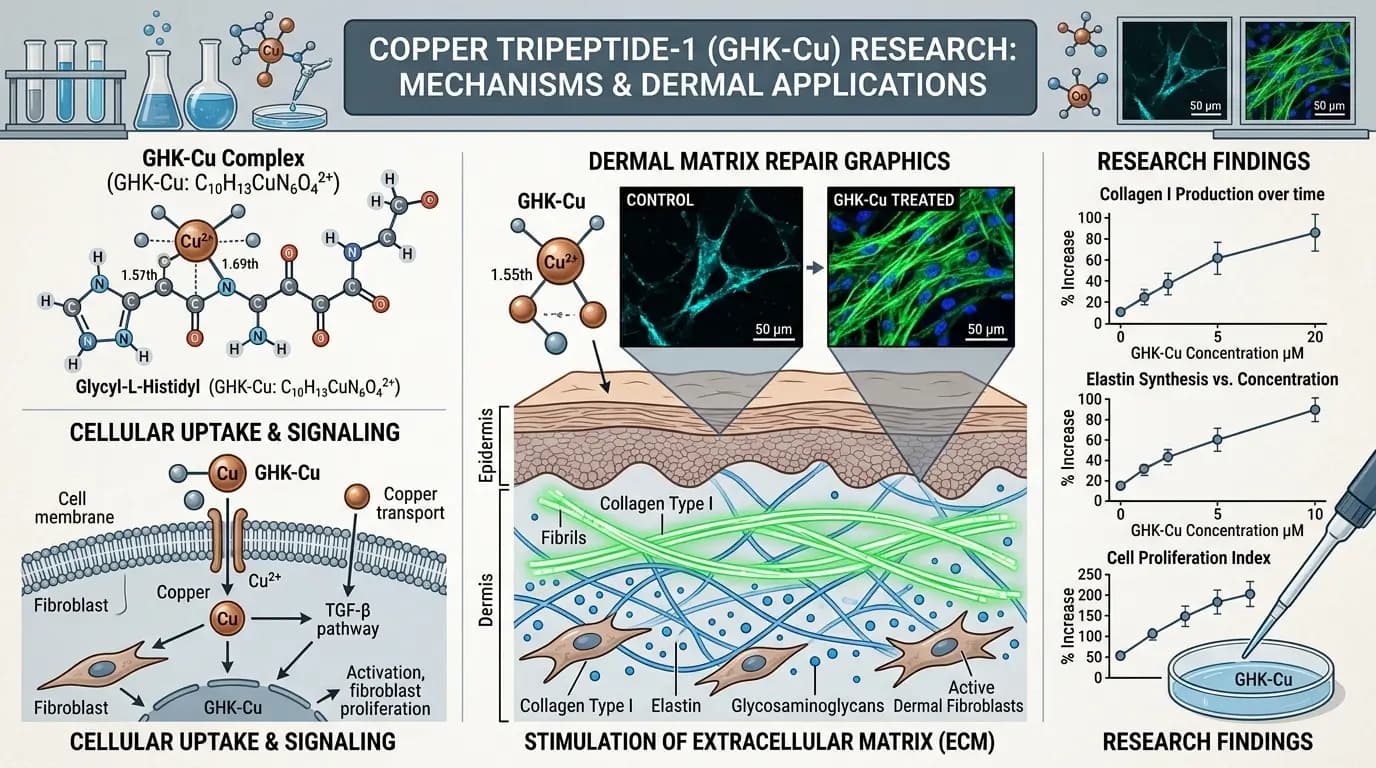
GHK-Cu benefits research has become a frequent topic in peptide science because this copper-binding tripeptide appears in many preclinical papers on tissue biology. For beginners, GHK-Cu benefits research can look confusing at first, since studies use different models, endpoints, and lab methods. For experienced readers, GHK-Cu benefits research still needs careful interpretation because findings from cell systems, ex vivo tissues, and animal models do not always align. This guide explains GHK-Cu benefits research in a clear, research-first way, with attention to methods, limitations, and evidence quality.
This article presents GHK-Cu benefits research as a laboratory topic only. The focus stays on model design, biomarker trends, and interpretation rules that help readers compare studies. You will see where GHK-Cu benefits research is strongest, where it is mixed, and where uncertainty remains high. You will also get a practical framework for reading GHK-Cu benefits research without overextending conclusions.
For context, researchers often group GHK-Cu benefits research into three large buckets: matrix biology, hair-follicle biology, and wound-focused model systems. However, each bucket contains multiple sub-models, and each sub-model has its own constraints. Therefore, this guide separates headline claims from underlying data patterns. In addition, it highlights reporting quality factors that make GHK-Cu benefits research more useful for lab teams and content reviewers.
What GHK-Cu Is and Why It Is Studied
GHK-Cu benefits research starts with chemistry. GHK is a tripeptide, glycyl-L-histidyl-L-lysine, and Cu refers to copper in a bound complex form. In many papers, GHK-Cu benefits research examines how this complex interacts with matrix turnover, cell signaling, and stress-response pathways in controlled model systems. Because these are model-based contexts, interpretation must remain model-bound.
Researchers often track GHK-Cu benefits research using endpoint panels rather than single markers. For example, one study may measure collagen-associated transcripts, while another measures vascular markers, inflammatory signals, or histology-based tissue features. Consequently, GHK-Cu benefits research should be read as a pattern of evidence across endpoints, not as one metric that settles the question.
The table below summarizes common technical descriptors used when reviewing GHK-Cu benefits research.
Parameter | Typical lab reporting focus | Why it matters in GHK-Cu benefits research |
|---|---|---|
Compound identity | Peptide sequence and copper complex notation | Confirms material identity before comparing papers |
Purity profile | Percentage purity and impurity disclosure | Supports comparability across batches |
Model type | In vitro, ex vivo, or animal model | Defines interpretation boundaries |
Endpoint class | Gene expression, protein signal, histology, imaging | Shapes what can be inferred |
Exposure window | Short pulse, repeated exposure, or longitudinal window | Influences signal direction and magnitude |
Statistical frame | Effect size, variance, threshold, replication count | Helps assess confidence in findings |
As a practical point, GHK-Cu benefits research can look stronger than it is when papers mix exploratory endpoints with confirmatory language. For that reason, readers should check whether studies predefine primary endpoints and whether replication details are clear. Meanwhile, narrative summaries should avoid turning exploratory observations into broad conclusions.
GHK-Cu Benefits Research in Skin and Matrix Biology
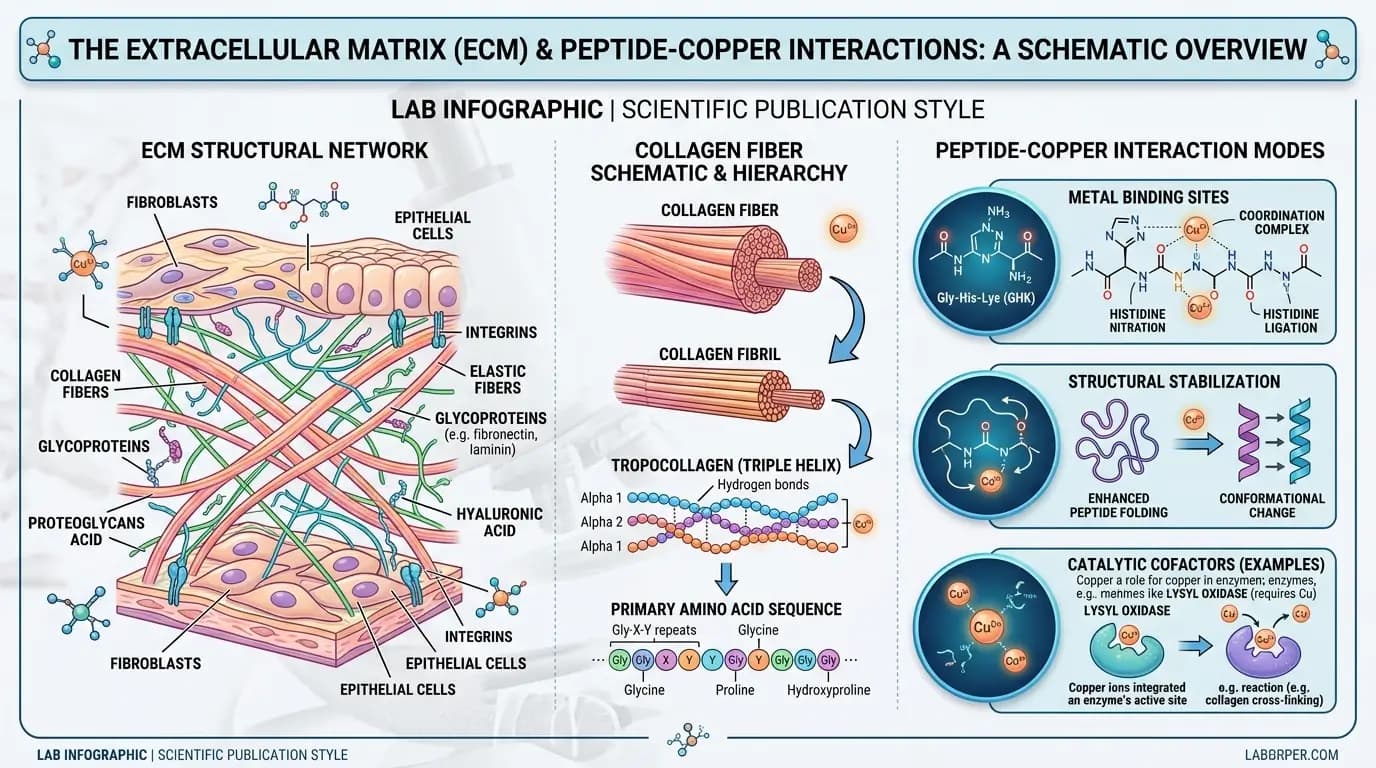
A major share of GHK-Cu benefits research examines extracellular matrix dynamics. Many studies report that matrix-related markers move in directions associated with remodeling activity. Yet, matrix biology is multi-layered, and signals can vary by cell source, assay format, and time point. Therefore, GHK-Cu benefits research in this area should be framed as context-dependent, not universal.
In cell-based systems, GHK-Cu benefits research often evaluates fibroblast-associated markers, matrix proteins, and related transcriptional profiles. Some datasets show increased expression of matrix-linked factors under specific conditions. However, other datasets show smaller shifts or mixed directionality depending on concentration and assay design. Because of this spread, cross-study comparison requires consistent attention to concentration range, media conditions, and normalization methods.
Another recurring theme in GHK-Cu benefits research is matrix turnover balance. Matrix renewal involves both synthesis-associated and degradation-associated pathways, so one-direction interpretation is usually incomplete. In addition, GHK-Cu benefits research in matrix contexts may include oxidative stress markers, inflammatory mediators, and signaling nodes that interact with matrix pathways. This interaction can complicate straightforward readouts.
For beginners, one helpful rule is simple: in GHK-Cu benefits research, stronger interpretation comes from converging evidence across transcript, protein, and morphology layers. If only one layer moves while others are unchanged, interpretation should stay cautious. For researchers, this means ranking papers by assay depth and replication quality, not by abstract language.
GHK-Cu Benefits Research in Hair Follicle Models
Hair-focused GHK-Cu benefits research usually studies follicle microenvironment biology, including signaling, matrix context, and stress-response pathways. The common challenge is model heterogeneity. Follicle biology differs across species, tissue sources, and culture designs. Consequently, GHK-Cu benefits research in this area should be mapped model by model before any broad summary.
Some GHK-Cu benefits research evaluates signaling pathways linked to inflammatory tone, oxidative stress handling, and local matrix behavior in follicle-adjacent systems. These findings can be informative for mechanism generation. Even so, mechanism generation is not the same as endpoint confirmation across independent labs. Therefore, readers should separate hypothesis-supporting observations from high-confidence evidence tiers.
In addition, GHK-Cu benefits research in hair-related systems may include endpoints such as follicle morphology scoring, marker staining, and selected growth-phase indicators. These endpoints can differ widely in robustness. For example, morphology grading without blinded assessment can introduce interpretation bias. Likewise, small sample sets can magnify apparent signal strength.
A useful interpretation framework is to classify GHK-Cu benefits research into three confidence bands: exploratory, intermediate, and replication-backed. Exploratory papers are valuable for ideas. Intermediate papers connect ideas to structured endpoints. Replication-backed papers, especially with transparent methods, offer stronger guidance for future lab design. This framework helps both beginners and advanced readers evaluate hair-focused GHK-Cu benefits research in a consistent way.
GHK-Cu Benefits Research in Wound Model Systems
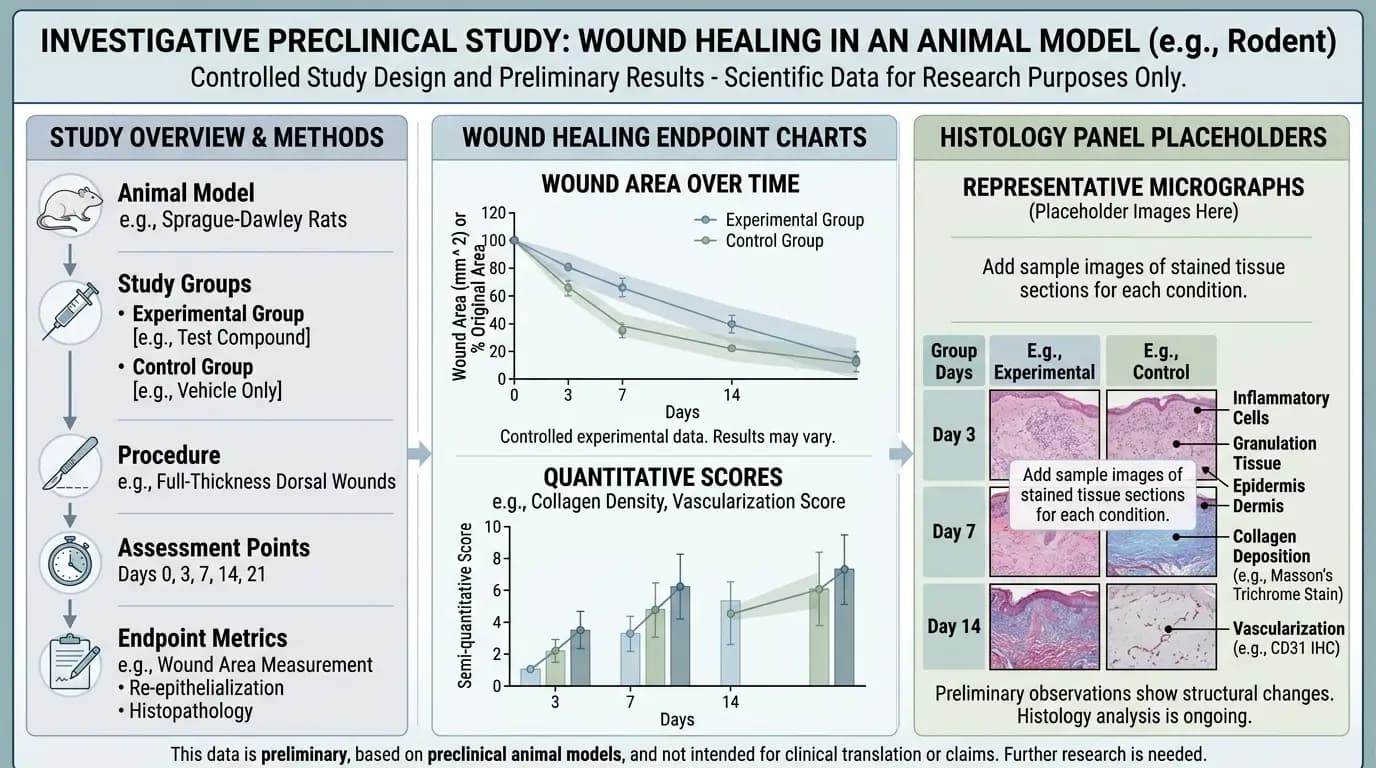
Wound-focused GHK-Cu benefits research is one of the most discussed areas, partly because model outcomes can be easier to visualize through imaging and histology. Still, visual clarity does not guarantee interpretive clarity. Wound model systems vary in injury type, timing, measurement windows, and tissue context. As a result, GHK-Cu benefits research in wound settings often shows mixed patterns across studies.
Some papers in GHK-Cu benefits research report directional shifts in selected wound-associated biomarkers. Others report neutral comparisons on key endpoints within specific model conditions. Mixed patterns like these are not unusual in preclinical science. Instead of forcing a single narrative, the better approach is to map which endpoints moved, under what model conditions, and with what statistical confidence.
For example, in certain wound-focused GHK-Cu benefits research, vascular or growth-factor staining outcomes may not differ meaningfully between test and control under defined conditions. That does not invalidate the full field. It means model context matters, and endpoint interpretation must remain specific to setup and constraints. Therefore, papers with neutral findings are as important as papers with positive directionality, because they improve evidence balance.
Researchers can improve comparability in GHK-Cu benefits research by standardizing model metadata reporting. This includes injury method details, timing of exposure, tissue sampling depth, imaging thresholds, and analysis pipelines. When these details are incomplete, cross-paper synthesis becomes fragile. When these details are explicit, evidence mapping becomes stronger and more useful.
Mechanisms Under Investigation in GHK-Cu Benefits Research
Mechanistic GHK-Cu benefits research often centers on signaling networks involved in matrix behavior, stress adaptation, and tissue-level remodeling. Mechanism papers can be informative because they identify plausible pathway interactions. However, a pathway signal is not automatically a broad functional conclusion. Therefore, mechanism claims should stay tied to measured endpoints and model boundaries.
Gene-expression datasets are also common in GHK-Cu benefits research. These datasets may show broad modulation across multiple genes linked to tissue biology. Yet, broad modulation alone does not determine endpoint direction in every model. In addition, transcriptional shifts may not map linearly to protein-level or tissue-level behavior. For this reason, mechanistic GHK-Cu benefits research is strongest when multi-layer validation is present.
Another point is temporal dynamics. Some pathway signals in GHK-Cu benefits research can differ between early and late windows. A marker may rise early and normalize later, or vice versa. If a paper reports only one time point, interpretation can miss this dynamic behavior. Therefore, studies with multi-timepoint designs provide richer context for mechanism-level reading.
From a practical standpoint, readers should ask three questions when reviewing mechanistic GHK-Cu benefits research:
Is the pathway signal consistent across independent assays?
Is the signal linked to a defined functional endpoint?
Is the interpretation limited to the tested model?
If any of these checks fails, conclusions should be phrased as preliminary.
Evidence Quality: How Strong Is the Current GHK-Cu Benefits Research?
Evidence quality is central to GHK-Cu benefits research because endpoint variation is high across the literature. A paper may look persuasive on first read, yet still carry design constraints that narrow interpretation. Therefore, quality review should be routine for every GHK-Cu benefits research summary.
First, sample structure matters. Small groups and narrow model diversity can produce unstable effect estimates. Second, endpoint selection matters. A panel with one favorable endpoint and several unchanged endpoints should be interpreted as mixed. Third, statistical transparency matters. Without clear variance reporting, threshold definitions, and replication counts, confidence drops.
Moreover, reproducibility signals are key in GHK-Cu benefits research. Independent confirmation across labs carries more weight than repeated analyses inside one pipeline. In addition, transparent method sections improve utility for other researchers who want to reproduce model conditions. Because of this, papers with detailed methods often contribute more to field progress than papers with broad claims but limited protocol disclosure.
For beginners, a simple quality ladder helps. Place each GHK-Cu benefits research paper on one of four rungs:
preliminary observation
structured exploratory evidence
model-level supportive evidence
replication-backed evidence
This ladder does not rank by excitement. Instead, it ranks by interpretive reliability.
How Beginners and Researchers Can Read GHK-Cu Benefits Research Better
Reading GHK-Cu benefits research well starts with intent clarity. If your goal is education, prioritize review papers and transparent methods. If your goal is lab planning, prioritize studies with clearly defined endpoints and replication detail. In either case, avoid overreading any single paper.
The next step is endpoint mapping. In GHK-Cu benefits research, endpoint mapping means writing down exactly what was measured, how it was measured, and when it was measured. This prevents category errors, such as treating a short-window marker shift as a long-window tissue conclusion. In addition, endpoint mapping helps distinguish biological signal from measurement noise.
Then, apply model hierarchy. A practical hierarchy for GHK-Cu benefits research is: in vitro signaling context, ex vivo tissue context, then animal model context. Each level answers different questions. Therefore, synthesis should combine levels carefully instead of blending them into one statement. This approach improves clarity for both content teams and technical readers.
Finally, document uncertainty directly. Strong GHK-Cu benefits research communication includes what is known, what is mixed, and what remains open. Clear uncertainty language does not weaken scientific writing. On the contrary, it improves trust and supports better decisions in research communication workflows.
Research Documentation Checklist for GHK-Cu Benefits Research
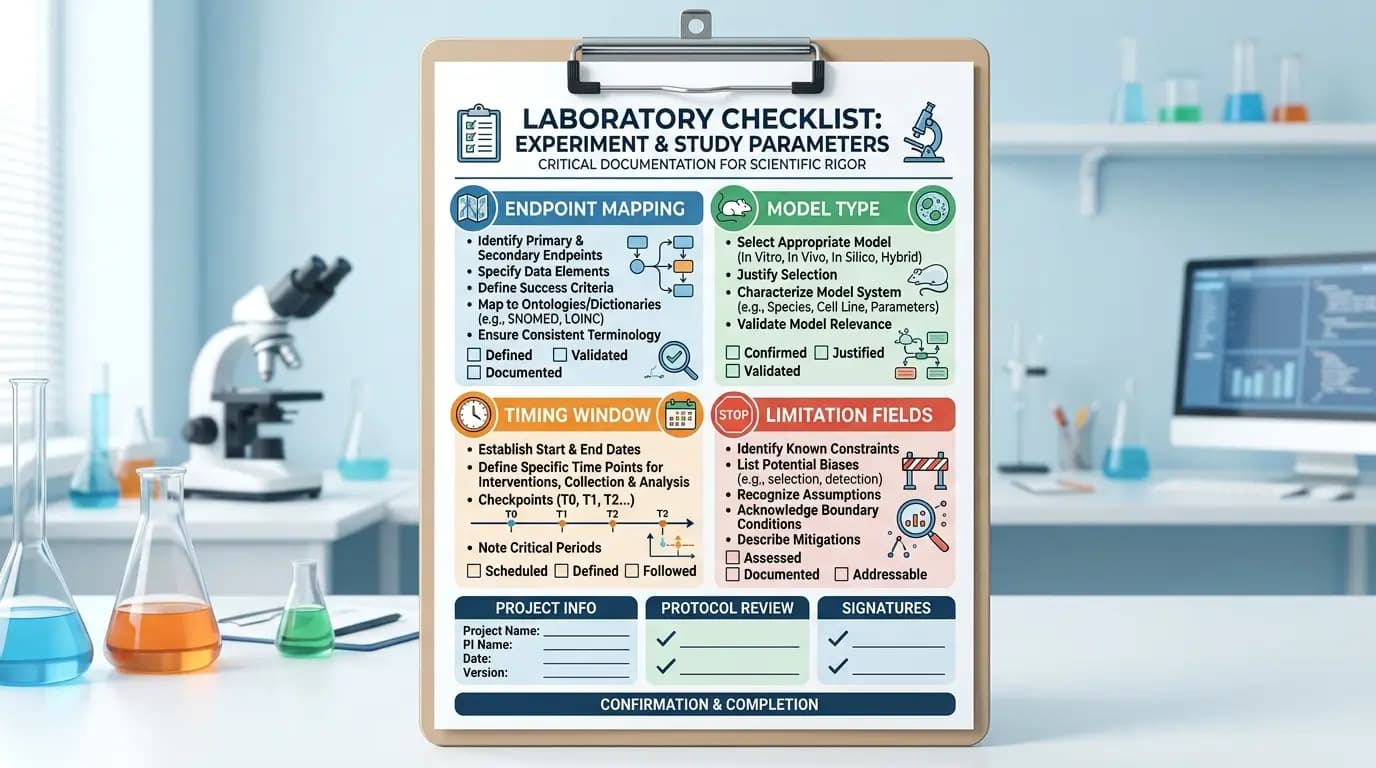
When preparing a blog, catalog note, or educational page on GHK-Cu benefits research, the checklist below improves quality and consistency.
Define model type clearly (in vitro, ex vivo, animal model)
State endpoint classes before discussing findings
Report timing windows and exposure context
Separate directional signals from confirmed replication patterns
Include neutral and mixed papers, not only favorable directionality
Note statistical and methodological limits in plain language
Use model-bound wording such as “in this model” or “under these conditions”
Keep conclusions aligned with measured endpoints only
In addition, include at least one small evidence map for readers who are new to GHK-Cu benefits research. This can be a mini-table, endpoint matrix, or model comparison grid. Structured presentation improves comprehension and lowers misinterpretation risk.
Conclusion
GHK-Cu benefits research is an active preclinical topic with meaningful scientific interest, especially in matrix biology, follicle-related model systems, and wound-focused designs. At the same time, GHK-Cu benefits research contains mixed endpoint patterns and model-specific variation, which means interpretation must remain disciplined. The strongest reading strategy is to combine model hierarchy, endpoint mapping, and evidence-quality review.
For beginners, the main takeaway is clarity: GHK-Cu benefits research is best understood as a spectrum of laboratory findings rather than one universal claim. For researchers, the next step is precision: prioritize transparent methods, multi-layer endpoints, and replication-centered interpretation. When these practices are applied, GHK-Cu benefits research becomes easier to evaluate, compare, and communicate in a scientifically responsible way.
For laboratory research use only. Not intended for use in humans or animals. Not intended to diagnose, treat, cure, or prevent any disease.
References
Pickart, L., & Margolina, A. “GHK Peptide as a Natural Modulator of Multiple Cellular Pathways in Skin Regeneration.” BioMed Research International (2015). https://ncbi.nlm.nih.gov/pmc/articles/PMC4508379/
Tenaud, I., et al. “In vivo stimulation of connective tissue accumulation by the tripeptide-copper complex glycyl-L-histidyl-L-lysine-Cu2+ in rat experimental wounds.” Journal of Clinical Investigation citations page. https://www.jci.org/articles/view/116842/citations/year/2009
Canapp, S. O., et al. “Effects of topical copper tripeptide complex on wound healing in an irradiated rat model.” Otolaryngology–Head and Neck Surgery (2013). PubMed: https://pubmed.ncbi.nlm.nih.gov/23744835/
Maquart, F. X., et al. “Effects of Copper Tripeptide on the Growth and Expression of Growth Factors by Normal and Irradiated Fibroblasts.” Archives of Facial Plastic Surgery listing page. https://www.liebertpub.com/doi/10.1001/archfaci.7.1.27