Bac water appears in research catalogs so often that many beginners assume every sterile water vial belongs to the same category. In label language, bac water means bacteriostatic water, a sterile water preparation that contains benzyl alcohol as a preservative. Official references also describe it as a container designed for repeated entries, which is the key detail behind storage rules and opened-vial dating.
That is why a single casual rule cannot answer the question “how long is bac water good for.” Unopened stock follows the manufacturer’s expiration date. After first entry, U.S. guidance for repeated-entry vials points to a 28-day window unless the label states another period, and that opened-vial window cannot go past the original expiration date.
This article explains bacteriostatic water for beginners and researchers in a simple, catalog-style way. It focuses on composition, bac water vs sterile water, storage, shelf life, and buying checks. It does not offer personal-use guidance.
What Is Bac Water?
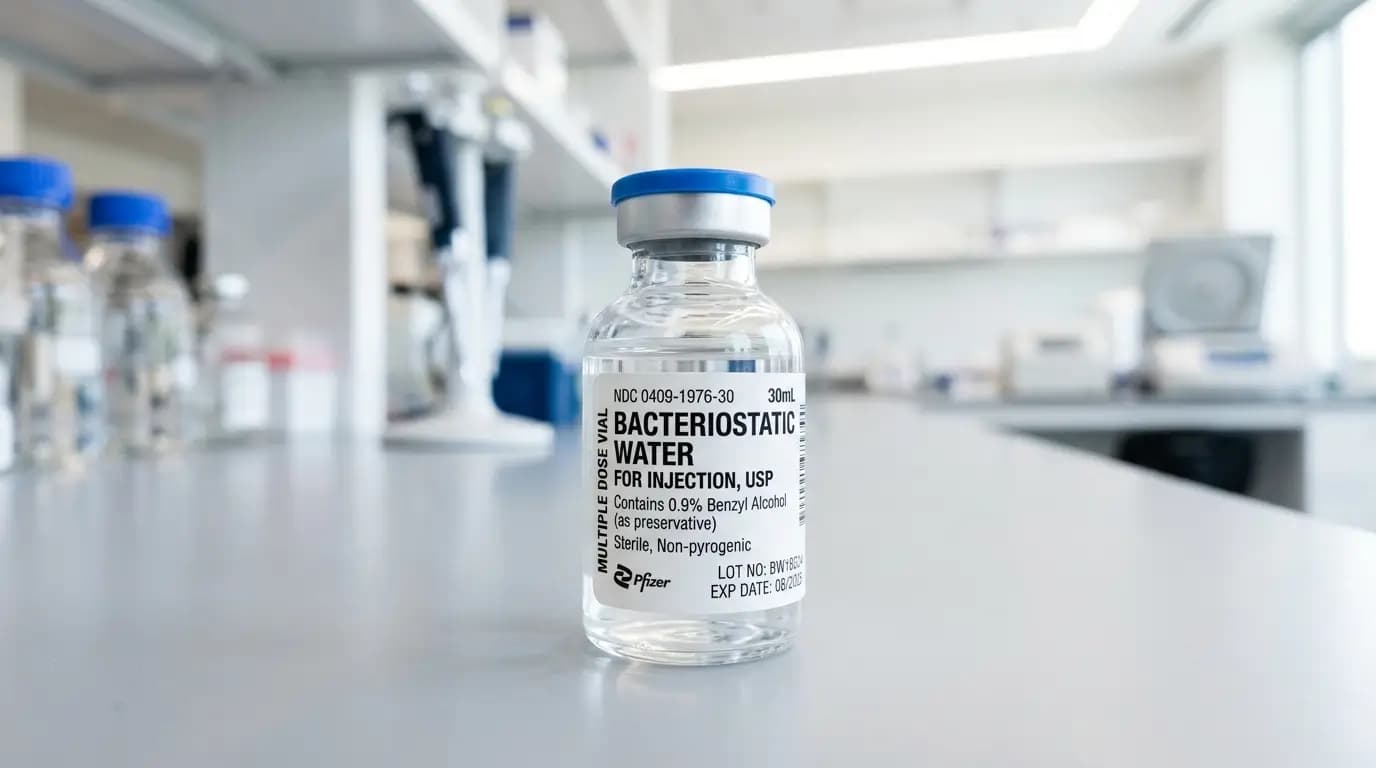
What “bac water” means
Bac water is the common shorthand for bacteriostatic water. DailyMed and Pfizer labeling describe bacteriostatic water as sterile, nonpyrogenic water with benzyl alcohol added as a bacteriostatic preservative. So, bac water is not just generic sterile water in a vial. It is a defined product class with its own label language, handling logic, and shelf-life controls.
For a beginner, the easiest way to think about bac water is to split the name into two parts. “Water” tells you the base material. “Bacteriostatic” tells you a preservative has been added to help limit bacterial growth inside the labeled container. However, that preservative does not remove the need for date control, inspection, or disciplined handling.
The basic composition of bac water
The formula behind bac water is simple. Official labeling states that bacteriostatic water contains water plus either 0.9 percent, equal to 9 mg/mL, or 1.1 percent, equal to 11 mg/mL, benzyl alcohol. The same references list a pH around 5.7, with a range of 4.5 to 7.0.
Because the composition looks simple, some buyers focus only on price or vial size. Yet the more useful details are the preservative concentration, the storage statement, the original expiration date, and the exact package type. Those details explain far more about a bac water listing than short marketing copy does.
Why benzyl alcohol is included
The CDC explains that vials intended for repeated entries typically contain an antimicrobial preservative to help limit bacterial growth. Bac water fits that category because its benzyl alcohol is there to support that preserved status. In other words, the preservative is the reason the product sits in a different handling class from preservative-free sterile water.
Still, the preservative has limits. The CDC notes that it does not affect viruses and does not provide complete protection against bacterial contamination. Therefore, bac water should always be managed with the same care you would expect from any documented laboratory material.
Bac Water in Laboratory Research
Why labs distinguish bac water from plain sterile water
Laboratories separate bac water from plain sterile water because the labels describe two different product types. Official sterile water labels state that sterile water contains no bacteriostat, no antimicrobial agent, and no added preservative. Those same label references describe sterile water as a single-entry product.
By contrast, manufacturers preserve bacteriostatic water for repeated entries under appropriate handling. That difference changes how staff date the vial after opening, store it on the shelf, and log it in inventory. Therefore, treating bac water and sterile water as interchangeable can create confusion long before someone opens the material.
Where bac water fits in routine handling
In routine lab handling, treat bac water as a preserved aqueous diluent that belongs inside a clear materials workflow. That means the team checks the label on arrival, verifies the expiration date, stores the vial in the listed range, and writes the opening date on the container when they first enter the vial.
A small lab does not need a complicated workflow. A simple receiving check, a storage location, and an opened-vial date can answer most day-to-day questions. In addition, this basic system makes it far easier to answer the common search query, how long is bac water good for, without relying on memory or guesswork.
Why labels and specifications matter
This material is one of those products where tiny label details carry real operational value. A good listing should make the preservative percentage, the storage range, and the container class easy to identify. When that information is visible, a buyer can tell whether the product matches the intended workflow before it reaches the bench.
This is also why label review matters more than broad blog claims. A post may mention bac water in general language, but the actual vial in front of you still controls its own storage and dating rules. Overall, the label is the first technical document that should guide every bac water decision.
Bac Water vs Sterile Water

The main difference in preservative content
The clearest bac water vs sterile water difference is preservative content. Bac water contains benzyl alcohol. Sterile water contains no bacteriostat, no antimicrobial agent, and no added preservative. That single distinction explains why people handle the two waters differently after opening.
For beginners, this is the comparison that matters most. Once you know whether preservative is present, you can usually predict the rest of the handling logic. Because of that, you should never evaluate bac water as “just another sterile water” listing.
Single-entry and repeated-entry container considerations
Sterile water labels describe a single-entry format, and some versions state that users should discard any unused amount after the first withdrawal. Bacteriostatic water labels, meanwhile, describe a vial intended for repeated entries. That difference shapes opened-vial dating from day one.
This does not mean one product is always preferable. Rather, each product fits a different packaging logic. For example, a workflow that requires preservative-free material will not be looking for bac water. In contrast, a workflow built around a preserved repeated-entry vial will read the bac water label first.
Quick comparison table
The table below summarizes the label-level differences most buyers want to see first.
Feature | Bac water | Sterile water |
|---|---|---|
Preservative | Benzyl alcohol present | No preservative |
Container logic | Repeated-entry vial | Single-entry vial |
Opened-vial timing | Date on first entry, usually 28 days unless label says otherwise | Unused portion discarded after first entry on relevant label versions |
Shelf reading | Track opening date and original expiry | Track original expiry and single-entry status |
These points come from official label language and vial-handling guidance, not from casual forum summaries.
How Long Is Bac Water Good For?
Unopened bac water and shelf life basics
Unopened bac water is good through the manufacturer’s expiration date when the vial remains in its original state and is stored under the listed conditions. The CDC states that unopened vials in this category should be discarded according to the manufacturer’s expiration date. So, the unopened answer to “how long is bac water good for” starts with the printed label, not with a generic internet estimate.
This label-first approach matters because packaging can vary. One product photo may look almost identical to another, yet the storage wording, package format, or original dating may differ. Therefore, unopened bac water should always be evaluated by its own container and carton details.
Opened bac water after first entry
Once the vial is first entered, the timing changes. The CDC states that an opened repeated-entry vial should be dated and discarded within 28 days unless the manufacturer states another period. The FDA also notes that 28 days is the assumed opened-vial period for this kind of container unless the label specifies something else.
That means the practical answer to “how long is bac water good for after opening” is usually up to 28 days, provided the original expiration date has not arrived earlier and the vial still meets visual and handling checks. This is a documentation rule as much as a storage rule. Once the opening date is missing, the status of the vial becomes much harder to defend.
What can shorten the usable window
The 28-day rule does not remove the need for inspection. The CDC explains that preservatives help limit bacterial growth but do not provide complete protection. Official bacteriostatic water labeling also says the solution should be clear and the seal intact.
As a result, the usable window can shorten when the seal is damaged, the solution is no longer clear, the storage range has not been followed, or the vial has been handled without consistent aseptic technique. For that reason, the shelf-life question always depends on both the date record and the current condition of the container.
Why label review still matters
A lot of confusion around this product comes from reading summaries without checking the product itself. Yet the vial label is still the deciding document. It tells you the storage range, the preservative status, and the original expiry. Meanwhile, CDC and FDA guidance tell you how to handle the opened-vial window when the product belongs to a repeated-entry class.
For a beginner lab, that is good news. You do not need a complex decision tree to answer how long bac water is good for. You need the vial, the opening date, the original expiration date, and the container’s present condition.
How to Store Bac Water Properly

Temperature, light, and storage conditions
Official labeling for bacteriostatic water lists storage at 20 to 25°C, which is controlled room temperature. So, unless the specific product says otherwise, bac water should be read as a room-temperature material with a defined label range.
That sounds straightforward, but it removes a common beginner mistake. Do not invent a storage rule for bac water from comments, screenshots, or random product blurbs. Instead, read the actual storage line on the container or carton and keep the vial within that range.
Stopper condition and container integrity
Storage is not only about temperature. Official labeling also states that the solution should be clear and the seal intact. In practice, this means container condition belongs in every storage check, not only in a final inspection.
A useful routine is simple: look at the vial before every entry, confirm the label remains readable, and confirm the opening date is present. If the seal looks compromised or the record is incomplete, the cleanest move is to remove the vial from active stock and follow internal lab policy.
What researchers should check before each entry
A short pre-check keeps bac water handling consistent:
Confirm the vial is bacteriostatic water and not preservative-free sterile water.
Confirm the original expiration date has not passed.
Confirm the opening date is written on the label.
Confirm the opened-vial window has not been exceeded.
Confirm the solution remains clear.
Confirm the seal and stopper area still look intact.
Confirm storage conditions match the label.
For beginners, this checklist is often enough to answer the shelf-life question on the spot. If every line passes, the vial remains within its documented window. If one line fails, the lab should follow its internal material-control rules rather than rely on assumption.
Common Mistakes and Misunderstandings
Bac water is not the same as plain sterile water
The biggest misunderstanding is also the simplest one. Bac water and sterile water are not the same item with two different names. One is preserved. The other is preservative-free. One fits repeated-entry handling. The other fits single-entry handling.
Because of that, the two products answer different workflow needs. In addition, they follow different logic after opening. So, a buyer who learns this one distinction early will avoid most basic catalog errors.
Repeated-entry status does not mean unlimited shelf life
Another frequent mistake is assuming the vial remains acceptable until it is empty. CDC and FDA guidance say otherwise. Once a repeated-entry vial is opened, a time limit applies, and that limit is usually 28 days unless the label states another period.
So, the right answer to “how long is bac water good for” is never “until the vial runs out.” The better answer is “until the documented opened-vial window ends, or sooner if the container no longer matches label condition.”
Why appearance changes matter
Official labeling says the solution should be clear and the seal intact. That is why appearance changes matter even before you finish checking dates. Cloudiness, visible particles, discoloration, or obvious container damage all point to a vial that no longer matches its labeled state.
Therefore, “it was stored on the shelf” is not enough documentation by itself. A vial’s status comes from a combination of the original expiry, the opening date, the storage range, and the current appearance of both the solution and the container.
What to Check Before Buying Bac Water

Preservative concentration
Before buying bac water, confirm the preservative concentration on the label or technical page. Official references for bacteriostatic water list benzyl alcohol at 0.9 percent or 1.1 percent. If a listing hides that basic detail, comparing one product to another becomes much harder than it should be.
Container size and packaging format
Next, check the vial size and the package logic. A serious bac water listing should make it clear that the product belongs to a repeated-entry category. This is one of the fastest ways to separate bac water from preservative-free sterile water while browsing supplier pages.
Lot details and documentation availability
Lot details matter because they support traceability. In a research purchasing workflow, batch-linked records make receiving checks, shelf rotation, and opened-vial tracking easier to manage. Moreover, they help answer later questions about which container was opened, when it was opened, and whether it was still inside its allowed window.
Label wording and storage guidance
Finally, read the storage line before ordering. A good listing should make the temperature range easy to find, and it should align with simple inspection points such as solution clarity and intact seals. When those basics are visible, the buyer can move from a vague keyword search to a controlled purchasing decision.
Conclusion
Bac water becomes much easier to understand once the label language is clear. It is bacteriostatic water with benzyl alcohol as a preservative, and it belongs to a repeated-entry product class. In contrast, plain sterile water is preservative-free and belongs to a single-entry class.
That is why “how long is bac water good for” has two answers. Unopened bac water follows the manufacturer’s expiration date. Opened bac water usually follows a 28-day window unless the label states another period, and that opened-vial window never extends beyond the original expiry.
For research buyers, the next step is straightforward. Read the bac water label, record the opening date, store the vial within the listed range, and inspect the container for clarity and seal integrity before each lab entry. That approach keeps shelf-life decisions simple, documented, and consistent.


